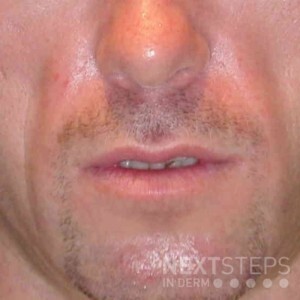
The correct answer is D. Poly-L-lactic acid (Sculptra).
Sculptra (called New-Fill outside of the US) is a biodegradable filler composed of poly-L-lactic acid, the same material used in absorbable sutures. It is biocompatible and nonallergenic and was approved by the FDA in August 2004 for the treatment of HIV-associated lipoatrophy.
Calcium hydroxyapatite and silicone are soft tissue fillers that are not FDA-approved for the treatment of lipoatrophy.
Botulinum toxin is used for chemodenervation.
Deoxycholic acid is FDA-approved for submental fat reduction, not lipoatrophy.
References:
Jones D. Volumizing the face with soft tissue fillers. Clin Plast Surg 2011; 38: pp. 379-390. PMID: 21824536
