Friday Pop Quiz 4/11/2025
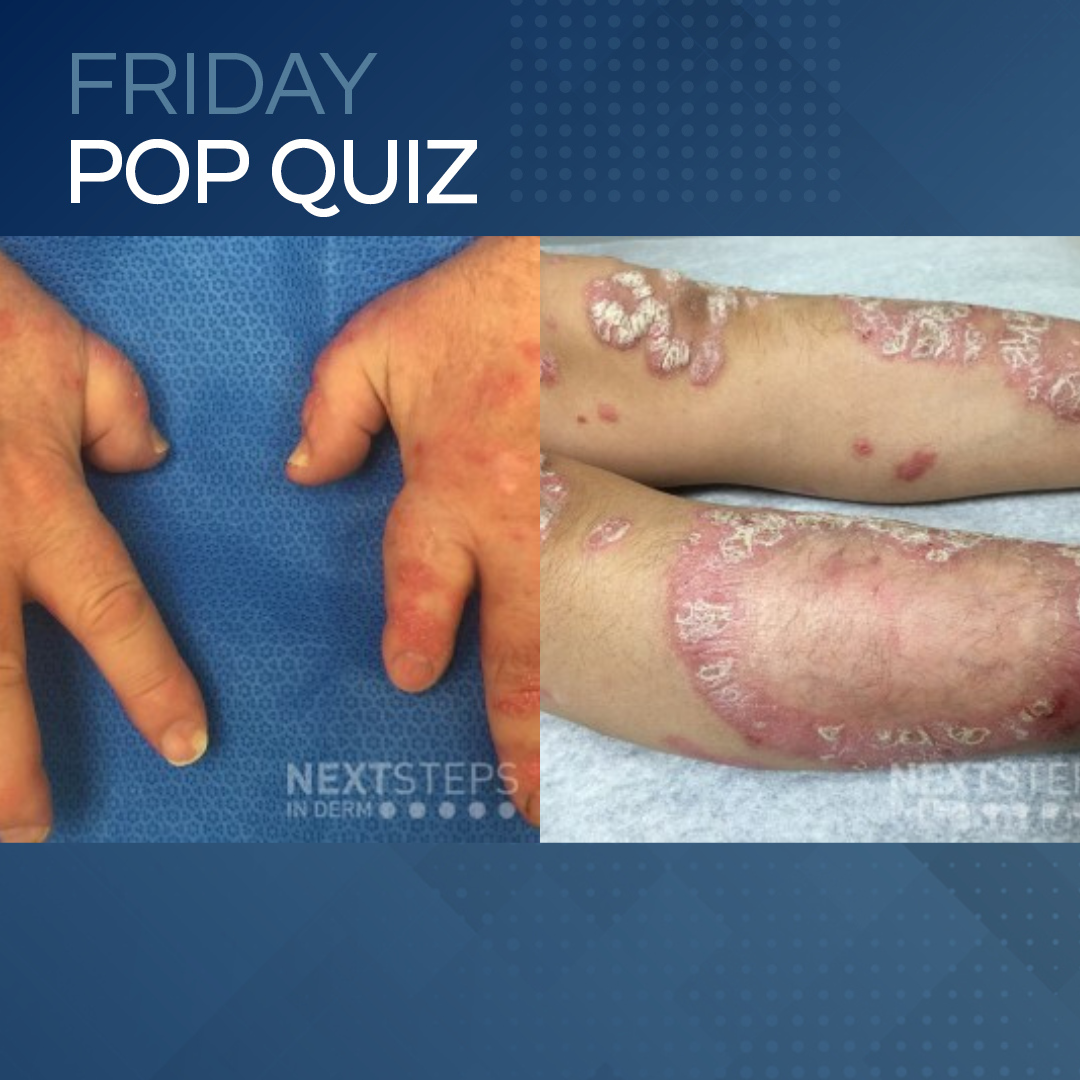 A 45-year-old patient with no prior skin problems presents with this new rash and review of system is positive for finger joint pain and swollen fingers. The pain is worse in the morning and improves as the day goes on. A thorough review of the patient’s medical history and labs were negative for HIV, hepatitis B and C, renal and he …
A 45-year-old patient with no prior skin problems presents with this new rash and review of system is positive for finger joint pain and swollen fingers. The pain is worse in the morning and improves as the day goes on. A thorough review of the patient’s medical history and labs were negative for HIV, hepatitis B and C, renal and he …
 A 45-year-old patient with no prior skin problems presents with this new rash and review of system is positive for finger joint pain and swollen fingers. The pain is worse in the morning and improves as the day goes on. A thorough review of the patient’s medical history and labs were negative for HIV, hepatitis B and C, renal and he …
A 45-year-old patient with no prior skin problems presents with this new rash and review of system is positive for finger joint pain and swollen fingers. The pain is worse in the morning and improves as the day goes on. A thorough review of the patient’s medical history and labs were negative for HIV, hepatitis B and C, renal and he … 

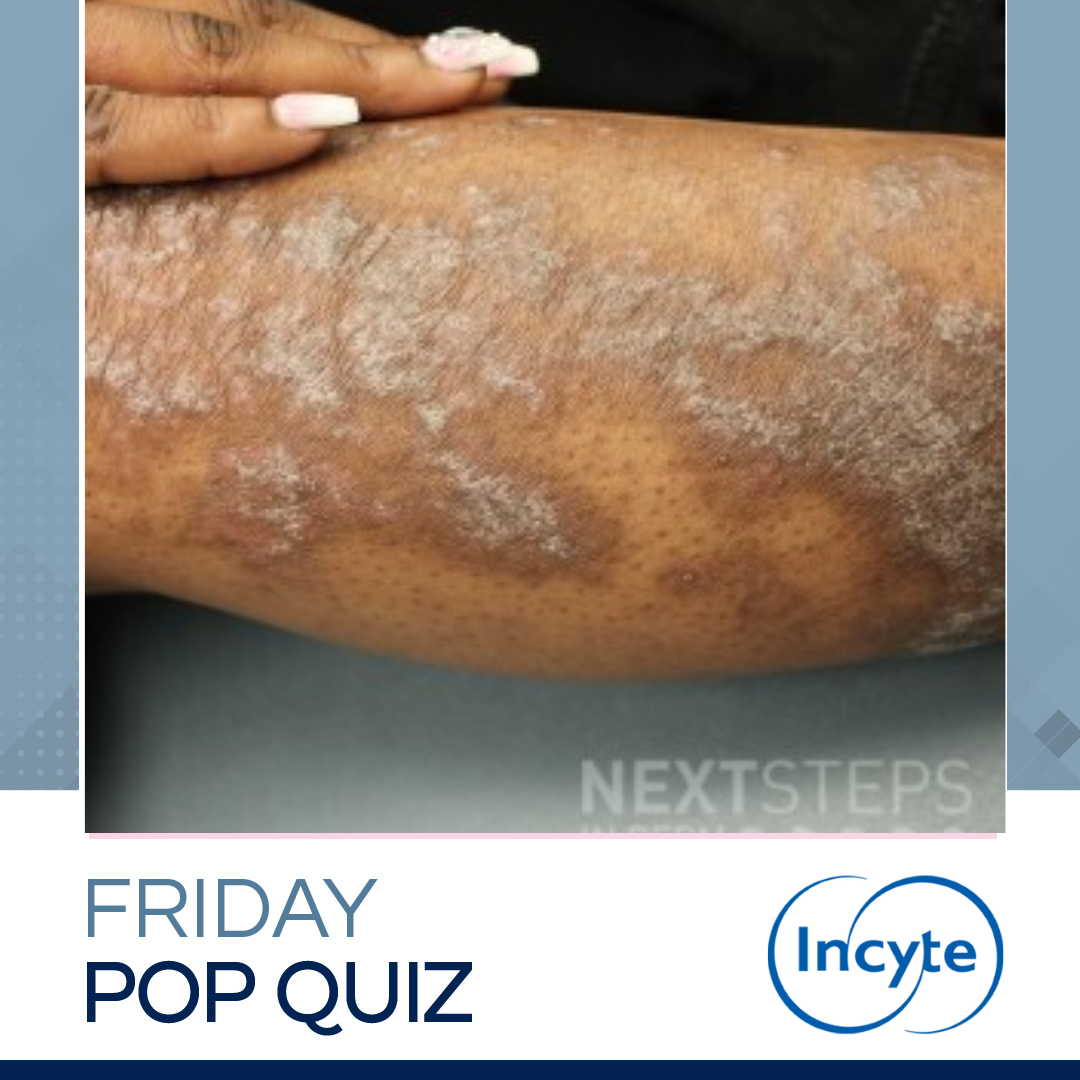 A 23-year-old female presents with itchy and irritated rash in the image. The rash began in her late teens. She has a family history of similar rash in her mother. Which histocompatibility antigen is most likely in this patient?
A. HLA-Cw2
B. HLA-Cw6
C. HLA-B51
D. HLA-B27
E. HLA-Bw35
To find out the correct answer and read the explanation, click here.
Brought to you by our bra …
A 23-year-old female presents with itchy and irritated rash in the image. The rash began in her late teens. She has a family history of similar rash in her mother. Which histocompatibility antigen is most likely in this patient?
A. HLA-Cw2
B. HLA-Cw6
C. HLA-B51
D. HLA-B27
E. HLA-Bw35
To find out the correct answer and read the explanation, click here.
Brought to you by our bra …  March into groundbreaking dermatological insights with the latest issue of the Journal of Drugs in Dermatology! Explore the prescribing habits of oral minoxidil for androgenetic alopecia, delve into a comprehensive review of botulinum toxin uses, and discover the effectiveness of microneedling for acne scars. Plus, unravel the uncertainties surrounding exosome therapy in androgenetic alopecia. Fro …
March into groundbreaking dermatological insights with the latest issue of the Journal of Drugs in Dermatology! Explore the prescribing habits of oral minoxidil for androgenetic alopecia, delve into a comprehensive review of botulinum toxin uses, and discover the effectiveness of microneedling for acne scars. Plus, unravel the uncertainties surrounding exosome therapy in androgenetic alopecia. Fro …  Next Steps in Derm, in partnership with Skin of Color Update, interviewed Dr. Shari Lipner, director of the Nail Division at the New York-Presbyterian Hospital/Weill Cornell Medical Center. Hear the results of Dr. Lipner’s retrospective study on time to diagnosis in patients with skin of color who have nail psoriasis. Find out why the clinical examination is especially important. Learn how derm …
Next Steps in Derm, in partnership with Skin of Color Update, interviewed Dr. Shari Lipner, director of the Nail Division at the New York-Presbyterian Hospital/Weill Cornell Medical Center. Hear the results of Dr. Lipner’s retrospective study on time to diagnosis in patients with skin of color who have nail psoriasis. Find out why the clinical examination is especially important. Learn how derm …  Bimekizumab (Bimzelx®) is a new injectable medication FDA approved for plaque psoriasis. It is the first and only FDA approved IL-17A and IL-17F inhibitor for this disease, but is also being used as an off-label option for other conditions including psoriatic arthritis and hidradenitis suppurativa. This Therapeutic Cheat Sheet will focus on the on and off label uses of bimekizumab.
Bimekizuma …
Bimekizumab (Bimzelx®) is a new injectable medication FDA approved for plaque psoriasis. It is the first and only FDA approved IL-17A and IL-17F inhibitor for this disease, but is also being used as an off-label option for other conditions including psoriatic arthritis and hidradenitis suppurativa. This Therapeutic Cheat Sheet will focus on the on and off label uses of bimekizumab.
Bimekizuma …