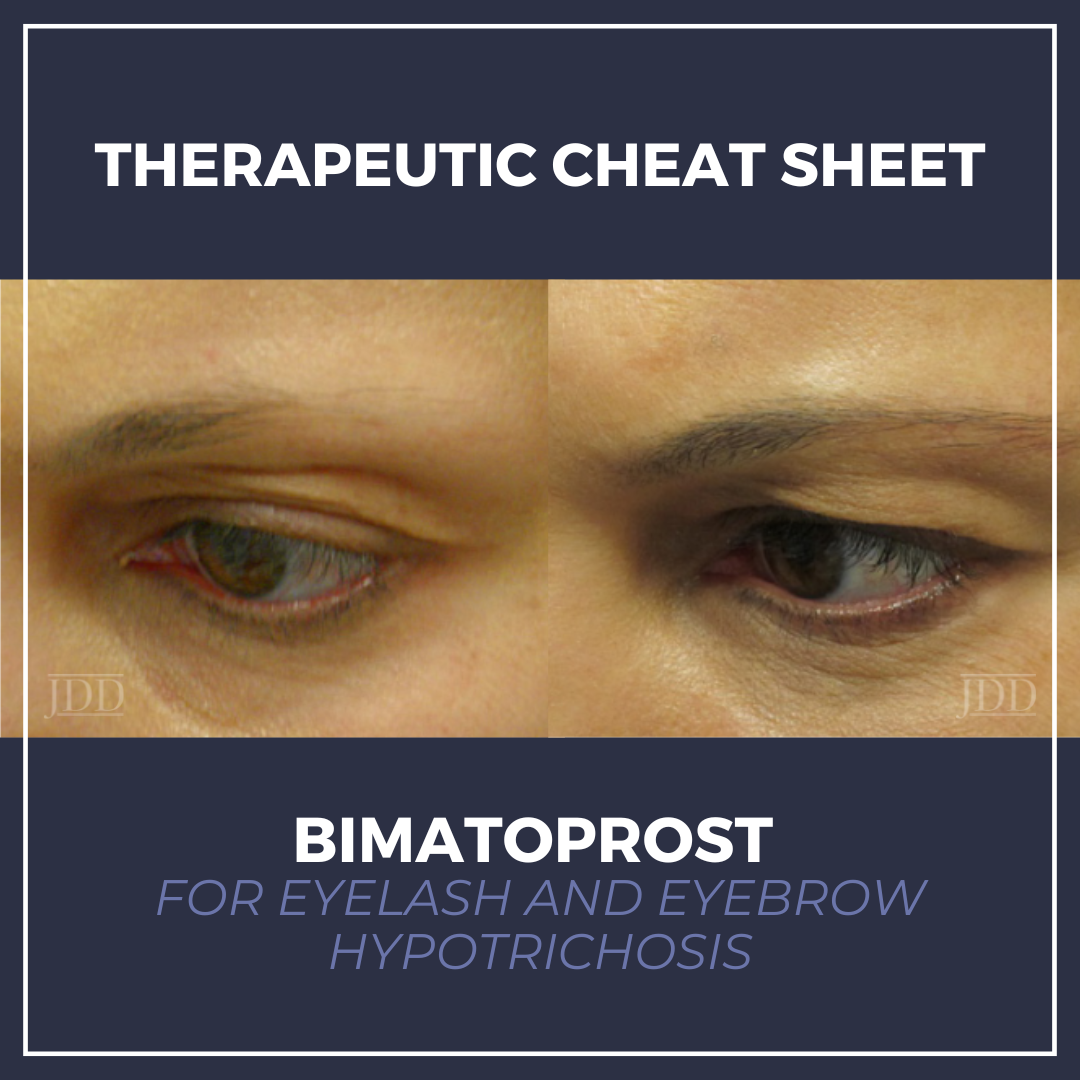
Bimatoprost is a prostaglandin analog originally developed for glaucoma that has become an important tool in dermatology for the treatment of eyelash and eyebrow hypotrichosis. By prolonging the anagen phase of hair growth, bimatoprost enhances lash length, thickness, and pigmentation. We continue our Therapeutic Cheat Sheet series with a closer look at bimatoprost.
Bimatoprost Therapeutic Cheat Sheet
Compiled by: Nidhi Shah, MD | Reviewed by: Adam Friedman, MD
TRADE NAMES1-2
-
- Latisse – 0.03% ophthalmic solution (FDA-approved for eyelash hypotrichosis)
- Lumigan – ophthalmic solution (for glaucoma/ocular hypertension)
MECHANISM OF ACTION1-3
-
- Bimatoprost is a synthetic prostamide F2a analog.
- The exact mechanism of action is not known but eyelash growth is thought to be stimulated by:
- Increasing the percentage of hairs in the anagen (growth) phase
- Prolonging duration of anagen phase
- Eyelashes have a higher proportion of hairs in the telogen phase which supports the effectiveness of bimatoprost for hypotrichosis of the eyelashes.
- Bimatoprost also stimulated melanogenesis which drives the periocular hyperpigmentation.
- When used intraocularly (Lumigan), lowers intraocular pressure (IOP) by increasing aqueous humor outflow
FDA APPROVED FOR1,2
-
- Latisse: Treatment of hypotrichosis of the eyelashes (increased length, thickness, and darkness).
- Lumigan: Reduction of elevated IOP in open-angle glaucoma or ocular hypertension.
OFF-LABEL DERMATOLOGIC USES3,4
-
- Eyebrow hypotrichosis
- Alopecia areata (eyelashes/eyebrows)
- Frontal fibrosing alopecia (eyebrow involvement)
- Chemotherapy-induced eyelash loss
- Adjunct in cicatricial alopecias affecting brows
- Androgenic alopecia
- Vitiligo
DOSING1
Eyelashes (FDA-approved use)
-
- Apply once nightly to clean, dry face. Using a sterile applicator, apply one drop to the upper eyelid margin only at the base of lashes and blot the excess solution.
- In the pivotal 16-week, multicenter, double-masked, randomized, vehicle-controlled clinical trial, significantly greater proportions of patients treated with bimatoprost achieved at least a 1-grade improvement in the 4-point Global Eyelash Assessment (GEA) scale compared to vehicle. Improvement was seen as early as Week 8 (50% vs 15%), increased at Week 12 (69% vs 20%), and reached 78% at Week 16 compared to 18% with vehicle. Digital image analysis at Week 16 demonstrated a mean increase in eyelash length of 1.4 mm (25% increase from baseline), a 106% increase in thickness/fullness, and an 18% increase in eyelash darkness. Upon discontinuation of treatment, eyelash growth gradually returns to baseline over several weeks to months.1
- Apply once nightly to clean, dry face. Using a sterile applicator, apply one drop to the upper eyelid margin only at the base of lashes and blot the excess solution.
Eyebrows (Off-label)
-
- Apply small amount with cotton-tipped applicator once nightly to affected eyebrow areas.
- Avoid runoff to forehead or unintended areas to prevent unwanted hypertrichosis.
WARNINGS AND PRECAUTIONS1
-
- May cause increased brown iris pigmentation, which is likely permanent.
- May cause eyelid skin hyperpigmentation; this is typically reversible after discontinuation.
- May cause periorbital changes including fat atrophy, deepening of the upper eyelid sulcus, and eyelid ptosis; these effects may be partially reversible.
- Unintended hypertrichosis may occur if the solution repeatedly contacts adjacent skin; excess medication should be blotted.
- May lower intraocular pressure (IOP); patients using prostaglandin analogs for glaucoma (e.g., Lumigan) should consult ophthalmology prior to use.
- Use with caution in patients with active intraocular inflammation (e.g., uveitis).
- Use with caution in aphakic patients, pseudophakic patients with a torn posterior lens capsule, or those at risk for macular edema.
- Contains benzalkonium chloride; soft contact lenses should be removed prior to application and reinserted after 15 minutes.
- Avoid contamination of the bottle tip and applicators to reduce risk of infection.
SIDE EFFECTS1
-
- Most common adverse effects (~3–4%) include eye pruritus, conjunctival hyperemia, ocular irritation, periorbital erythema, and skin hyperpigmentation.
- Rare side effects including blurred vision, eyelid edema, hordeolum, increased lacrimation, trichiasis, eyelash growth abnormalities, madarosis and trichorrhexis have been reported.
- Periorbital fat atrophy and eyelid sulcus deepening have been observed in postmarketing reports.
CONTRAINDICATIONS1
-
- Hypersensitivity to bimatoprost or formulation components.
DRUG INTERACTIONS1
-
- Caution with concurrent prostaglandin analogs used for IOP lowering (e.g., Lumigan).
- No major systemic drug interactions reported.
PREGNANCY & BREASTFEEDING1
-
- No adequate human studies with the use of bimatoprost in pregnant women. However, animal studies with oral systemic exposure at high doses showed abortion, early delivery, and reduced fetal weight.
- It is unknown if bimatoprost is excreted in human milk.
MONITORING1
-
- None
- Consider ophthalmology referral if patient has a history of glaucoma, ocular surgery, or new visual symptoms.

FURTHER READING
If you would like to learn more about Bimatoprost, check out the following articles in the Journal of Drugs in Dermatology:
Topical Bimatoprost for the Treatment of Eyebrow Hypotrichosis
Schweiger E, Pinchover L, Bernsterin R
Topical bimatoprost was FDA approved in December of 2008 for the treatment of eyelash hypotrichosis. Since its approval, some physicians have advocated the use of bimatoprost “off label” for hair growth in other areas, such as the scalp or eyebrows, but there has yet to be published scientific evidence to support this use. We report one of the first cases of significant eyebrow hair growth in a patient after use of topical bimatoprost for eyebrow hypotrichosis.
J Drugs Dermatol. 2012;11(1):106-108.
Bimatoprost 0.03% Solution for the Treatment of Nonfacial Vitiligo
Pearl E. Grimes MD
BACKGROUND: Topical prostaglandin E2 has shown efficacy in patients with localized, stable vitiligo. Bimatoprost is a synthetic prostamide (prostaglandin-ethanolamides) F2a analog. Bimatoprost 0.03% ophthalmic solution showed efficacy in the treatment of vitiligo in one small study.
OBJECTIVE: To assess the efficacy and safety of bimatoprost 0.03% alone and in combination with a topical steroid (mometasone) compared with mometasone alone in patients with nonsegmental vitiligo on nonfacial areas in a proof-of-concept study.
METHODS: This randomized, double-blind, controlled study was conducted over a 20-week treatment period. Patients were randomized to 1 of 3 treatment groups: bimatoprost monotherapy (n=11), bimatoprost plus mometasone (n=10), and mometasone plus placebo (n=11). The primary outcome was global response at week 20, based on an investigator’s assessment of improvement score of at least 5 (at least 50%–75% improvement from baseline) on an 8-point scale (0=worse; 7=cleared). Other outcomes included global response at other visits, response by anatomic site, change from baseline lesion severity (overall and by site), and safety.
RESULTS: Because of a lack of response observed for the primary end point, a post hoc analysis with a less stringent definition of response (score of ≥4 [25%–50% improvement]) was conducted. In this analysis, 46% of the bimatoprost plus mometasone group responded overall compared with 18% in the bimatoprost monotherapy group, and no patients in the mometasone plus placebo group. Greater response rates were observed in both bimatoprost groups compared with the mometasone plus placebo group starting at week 12. There were no differences among groups in signs and symptoms of irritation.
CONCLUSIONS: Bimatoprost alone or with mometasone provided greater repigmentation than treatment with mometasone alone. Larger studies that also assess facial lesions are warranted.
J Drugs Dermatol. 2016;15(6):703-710.
REFERENCES
-
- Allergan USA, Inc. Latisse (bimatoprost ophthalmic solution) 0.03% [prescribing information]. Revised August 2021. https://www.fda.gov/drugsatfda
- Allergan USA, Inc. LUMIGAN® (bimatoprost ophthalmic solution) 0.03% [prescribing information]. Revised August 2010. https://www.fda.gov/drugsatfda
- Jha AK, Sarkar R, Udayan UK, Roy PK, Jha AK, Chaudhary RKP. Bimatoprost in Dermatology. Indian Dermatol Online J. 2018;9(3):224-228. doi:10.4103/idoj.IDOJ_62_16
- Grimes PE. Bimatoprost 0.03% Solution for the Treatment of Nonfacial Vitiligo. J Drugs Dermatol. 2016;15(6):703-710.