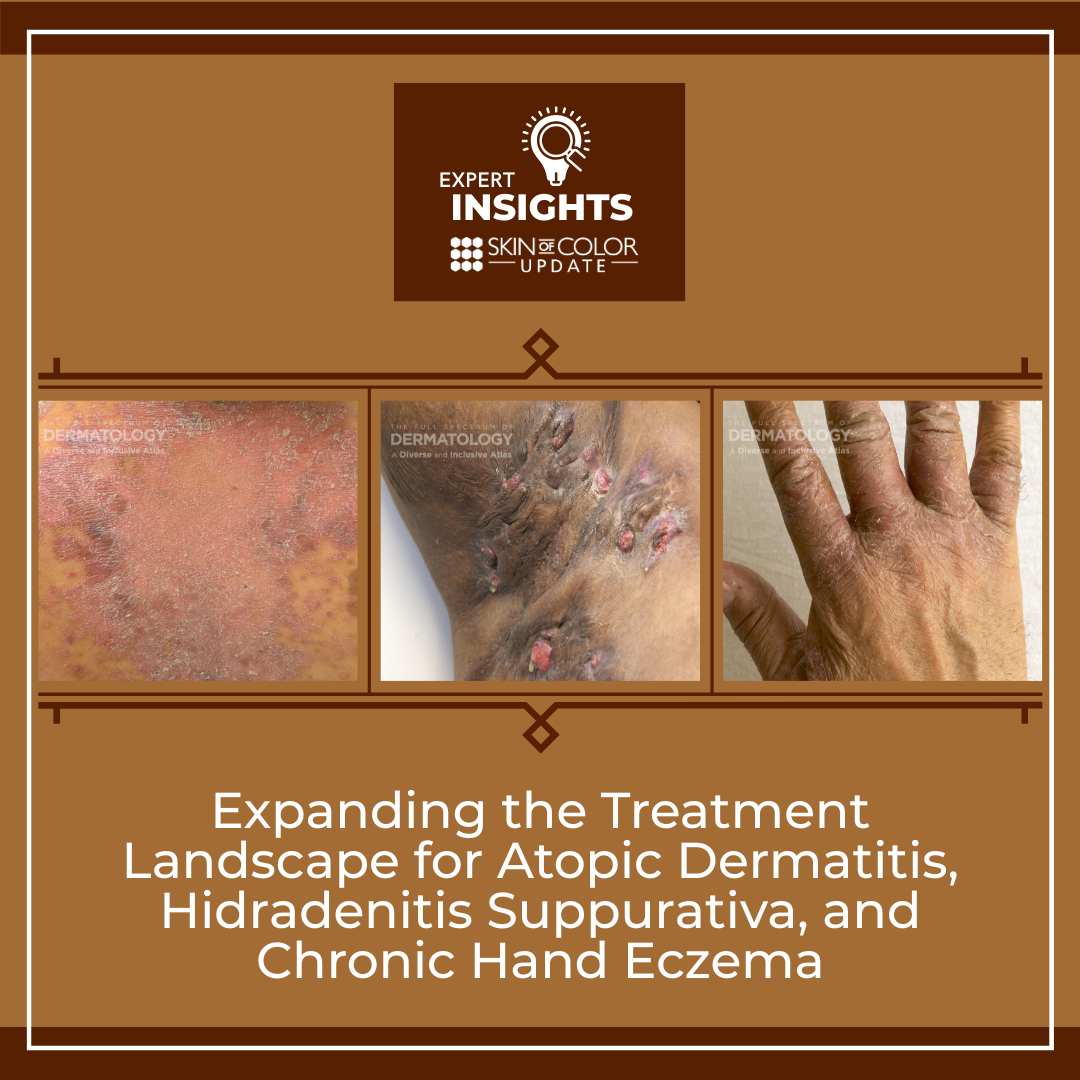
The past 18 months has seen an unprecedented wave of FDA approvals in dermatology, significantly expanding the therapeutic options for treatment of inflammatory skin diseases. At the 2025 Skin Of Color Update in New York City, conference co-chair Dr. Andrew Alexis highlighted some of these new medications and new indications for others. Importantly, many of these pivotal clinical trials included sizable cohorts of patients with skin of color, allowing for meaningful subpopulation analyses and new insights into treatment efficacy across diverse patient populations.
New Indications for Non-steroidal Topicals
Tapinarof is an aryl hydrocarbon receptor agonist with anti-inflammatory and skin barrier modulating activity. In December 2024, the FDA approved tapinarof 1% cream for atopic dermatitis for adults and pediatric patients 2 years and older. the ADORING 1 and 2 phase 3 clinical trials enrolled a large and racially/phototype-diverse cohort with substantial representation of patients with skin of color. Subgroup analyses showed comparable improvements in atopic dermatitis severity and response rates among Black/African American, Asian, and other racially diverse participants, as well as across all Fitzpatrick skin types. Importantly, treatment benefits in reducing disease burden and improving skin clearance were maintained without new safety concerns.
The PDE-4 inhibitor roflumilast was previously approved for seborrheic dermatitis. In May 2025, the FDA approved roflumilast topical foam 0.3% foam for once-daily treatment of plaque psoriasis of the scalp and body in adults and adolescents ≥12 years. Approval was based on two randomized, vehicle-controlled trials (including the phase 3 ARRECTOR study), which showed significantly clearance lesions over 8 weeks. Patients experienced rapid itch reduction, with improvements evident within 24 hours and sustained through the trial. The foam was well tolerated, with low discontinuation rates and the most common adverse events being headache, diarrhea, nausea, and nasopharyngitis.
The topical JAK inhibitor ruxolitinib was previously approved for vitiligo and atopic dermatitis in adult and adolescent patients aged 12 years and older. In September 2025, the FDA extended the approval of ruxolitinib 1.5% cream for patients with mild-to-moderate atopic dermatitis down to 2 years of age. Approval was based on the TRuE-AD3 clinical trial in which ruxolitinib showed a significant improvement in investigator global assessment, EASI75, and itch scores compared with vehicle. There were no new safety signals in this study, and most common adverse events were upper respiratory tract infections.
A New Treatment for Chronic Hand Eczema
In July 2025, the FDA approved twice daily application of the JAK inhibitor delgocitinib topical cream as the first treatment specifically indicated for moderate-to-severe chronic hand eczema. Approval was based on the phase 3 DELTA-1/2 trials showing significant improvement in investigator assessment and hand eczema severity assessment scores compared with vehicle at 16 weeks. Long-term extension data showed delgocitinib treatment was well-tolerated and continued to be efficacious in maintaining disease control up to 52 weeks.
Lebrikizumab for Atopic Dermatitis in Patients with Skin of Color
Approval of the anti-IL13 monoclonal antibody for the treatment of atopic dermatitis was announced in 2024. In the phase 3b open-label ADmirable trial, lebrikizumab showed robust efficacy and good safety over 24 weeks in moderate-to-severe atopic dermatitis in patients with Fitzpatrick skin types IV-VI. In this study, investigators used the new PCDA-Derm scale to quantify changes in post-inflammatory pigmentary alteration in these patients. They found 64.4% of patients wtih baseline hyperpigmentation showed improvement at week 24. This represents a clinically meaningful outcome in ameliorating a highly bothersome sequela of atopic dermatitis in patients with skin of color.
Nemolizumab for Atopic Dermatitis
The ant-IL31 receptor alpha monoclonal antibody nemolizumab was previously approved for adult patients with prurigo nodularis, in which IL31 is a major driver in the pathogenesis of chronic itch. In December 2024, the FDA approved nemolizumab for adolescents and adults aged 12 years and older with moderate-to-severe atopic dermatitis. Approval was supported by the phase 3 ARCADA 1/2 trials in which nemolizumab led to significant improvement in investigator global assessment and EASI75 over 16 weeks compared with placebo. Patients in this study were permitted concomitant use of topical steroids in both groups. Additionally, patients were allowed to switch from every-4-week dosing to every-8-week dosing if symptoms were well-controlled at the 16-week timepoint. There were infrequent cases of asthma and atopic dermatitis exaerbation in these studies.
Based on these approvals, Dr. Alexis highlighted the recent update on guidelines for care and management of atopic dermatitis in adults, which includes strong recommendations for new topical treatments, biologics, and JAK inhibitors in patients with moderate-to-severe disease.
Bimekizumab for Hidradenitis Suppurativa
In November 2024, the FDA approved the anti-IL17A/F monoclonal antibody bimekizumab for treatment of adults with moderate-to-severe hidradenitis suppurativa (HS). Data from the phase 3 BE HEARD I and II trials showed a significantly greater proportion of patients achieving HiSCR50 (≥50% reduction in abscesses and inflammatory nodules without worsening of abscesses or draining tunnels) with bimekizumab versus placebo at week 16, with this effect maintained at week 48. Safety findings included increased cases of candidasis and other fungal infections. There were a few cases of suicidal ideation (1.9% in bimikizemumab-treated patients vs. 0.7% in patients receiving placebo) in the first 16-weeks of the placebo-controlled trials.
Two New Indications for Dupilumab
In April 2025, FDA approved dupilumab for adults and adolescents aged 12 years and older with chronic spontaneous urticaria (CSU) refractory to antihistamine therapy. The LIBERTY-CSU CUPID trials demonstrated significant improvement in itch and UAS7 (number of urticarial wheals over a 7-day period) at 24 weeks compared with placebo.
In June 2025, FDA approved dupilumab as the first and only targeted therapy for adult patients with bullous pemphigoid (BP). The LIBERTY-BP ADEPT trial demonstrated significantly improved disease control, itch reduction, and decreased corticosteroid needs compared with placebo. There were no new safety signals compared with previous clinical trials involving dupilumab.
References
-
- Alexis AF, Kircik L, Chovatiya R, Rice ZP, Soong W, Bhutani T, Brown PM, Piscitelli SC, Rubenstein DS, Tallman AM, Armstrong AW. Tapinarof Cream for Adults and Children with Atopic Dermatitis-Efficacy by Race and Fitzpatrick Skin Type in Two Phase 3 Randomized Clinical Trials. Dermatol Ther (Heidelb). 2025 Sep;15(9):2667-2682. doi: 10.1007/s13555-025-01489-w. Epub 2025 Jul 22. PMID: 40696240; PMCID: PMC12354664.
- Gooderham MJ, Alonso-Llamazares J, Bagel J, et al. Roflumilast Foam, 0.3%, for Psoriasis of the Scalp and Body: The ARRECTOR Phase 3 Randomized Clinical Trial. JAMA Dermatol. 2025;161(7):698-706. doi:10.1001/jamadermatol.2025.1136
- Eichenfield LF, Stein Gold LF, Simpson EL, et al. Efficacy and safety of ruxolitinib cream in children aged 2 to 11 years with atopic dermatitis: Results from TRuE-AD3, a phase 3, randomized double-blind study. J Am Acad Dermatol. 2025;93(3):689-698. doi:10.1016/j.jaad.2025.05.1385
- Bissonnette R, Warren RB, Pinter A, et al. Efficacy and safety of delgocitinib cream in adults with moderate to severe chronic hand eczema (DELTA 1 and DELTA 2): results from multicentre, randomised, controlled, double-blind, phase 3 trials. Lancet. 2024;404(10451):461-473. doi:10.1016/S0140-6736(24)01027-4
- Alexis A, Moiin A, Waibel J, et al. Efficacy and Safety of Lebrikizumab in Adult and Adolescent Patients with Skin of Color and Moderate-to-Severe Atopic Dermatitis: Results from the Phase IIIb, Open-Label ADmirable Study. Am J Clin Dermatol. 2025;26(5):803-817. doi:10.1007/s40257-025-00970-8
- Silverberg JI, Wollenberg A, Reich A, et al. Nemolizumab with concomitant topical therapy in adolescents and adults with moderate-to-severe atopic dermatitis (ARCADIA 1 and ARCADIA 2): results from two replicate, double-blind, randomised controlled phase 3 trials. Lancet. 2024;404(10451):445-460. doi:10.1016/S0140-6736(24)01203-0
- Kimball AB, Jemec GBE, Sayed CJ, et al. Efficacy and safety of bimekizumab in patients with moderate-to-severe hidradenitis suppurativa (BE HEARD I and BE HEARD II): two 48-week, randomised, double-blind, placebo-controlled, multicentre phase 3 trials. Lancet. 2024;403(10443):2504-2519. doi:10.1016/S0140-6736(24)00101-6
- Maurer M, Casale TB, Saini SS, et al. Dupilumab in patients with chronic spontaneous urticaria (LIBERTY-CSU CUPID): Two randomized, double-blind, placebo-controlled, phase 3 trials. J Allergy Clin Immunol. 2024;154(1):184-194. doi:10.1016/j.jaci.2024.01.028
- Dupixent late-breaking positive pivotal data in bullous pemphigoid presented at AAD. Sanofi. March 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-03-08-18-00-00-3039375
This information was presented at the 2025 Skin of Color Update conference by Andrew F. Alexis, MD, MPH, FAAD. The above highlights from this lecture were written and compiled by Riyad N.H. Seervai, MD, PhD.