In part 1 of this 3-part series, we discussed the basics of moisturizer product design, including the key components that differentiate moisturizers from other product formats, and the benefits and uses of common ingredients. In part 2, we continue looking at other classes of ingredients, moisturization efficacy of creams versus lotions, and functional ingredients such as Niacinamide (Vitamin B3) and Alpha Hydroxy Acid (AHA), and PPARs just to name a few.
If you missed part 1 of this series, you can find it here.
Emulsifiers and Polymers
Moisturizers are usually emulsions or kinetically stabilized colloidal suspensions of two immiscible liquids meaning there is no appreciable phase separation and the in-use experience remains consistent over the product’s usable lifetime. Emulsions require the use of emulsifiers for stability. Emulsifiers can range from simple monomeric surfactants to much larger polymeric surfactants, particles, and lamellar liquid crystal aggregates.
An interesting property of emulsifiers is that they usually have long carbon chains like skin lipids, which makes it possible for an emulsifier to also impart skin benefits. In fact, the closer the chemistry of the emulsifier is to skin lipids, the more skin benefit can be imparted. Long chain emulsifiers, identified on an ingredient label as “palmitic” or “cetyl” (16 carbons or C16), “stearic” or “stearyl” (C18), and “behenic” or “behenyl” (C22), are compatible with and beneficial to skin, help maintain emulsion stability, and are not irritating. Stearic acid, a fatty acid with a C18 chain and carboxylic acid head group, is a good example of an emulsifier with skin benefits that Unilever uses extensively. Fatty acids are an integral component of the lipid matrix, and topically applied stearic acid has been demonstrated to incorporate into the lipid matrix improving the quality of the stratum corneum.11, 12
Another class of materials used to improve the stability, impact thickness and texture, and modify sensory feel of moisturizers is polymers. These could include synthetic polyacrylate type polymers or natural ones like starch (a polysaccharide-based polymer). The feel and look of emulsions changes dramatically depending upon the polymer and emulsifier used. For example, culinary enthusiasts are aware that using a starch versus gelatin results in radically different textures. Similarly, in cosmetics, availability of novel polymers and emulsifiers have provided the formulator with a unique ability to modulate the feel and texture of moisturizers independent of its efficacy.
Moisturization Efficacy: Cream or Lotion?
It is a widely held belief that creams are more moisturizing than lotions, and hence more effective in treating dry skin, for example. While this may have been true when such consumer products first appeared in the market, this is not the case anymore. Today, the difference between creams and lotions is largely related to the types of moisturizers and the consumer sensory expectations. Creams tend to be thicker; however, this can be easily modified by using the right combinations of polymers and emulsifiers. Creams can contain more oils than lotions as in the case of emollient creams, and lotions can contain more humectants, for example, Vaseline™ Intensive Care Deep Moisture Jelly Cream and Vaseline™ Intensive Care Advanced Repair Unscented (See Figure 1 in part 1 of this series),respectively. However, this is not always the case. In determining moisturization efficacy, the total level and combination of moisturizers in the product is the most important factor. This is clearly demonstrated in Figure 7, where hydration scores from a 3-week moisturization efficacy study with a 2-week regression phase were compared across different marketed cosmetic therapeutic creams (represented by dashed lines) and lotions (represented by solid lines). There was no direct relationship found between the format and efficacy, in fact, the most efficacious product in the study was a lotion. The entirety of the formulation is responsible for the product efficacy, but this data does suggest that glycerin and total moisturizer levels is a better indicator of performance than product format.
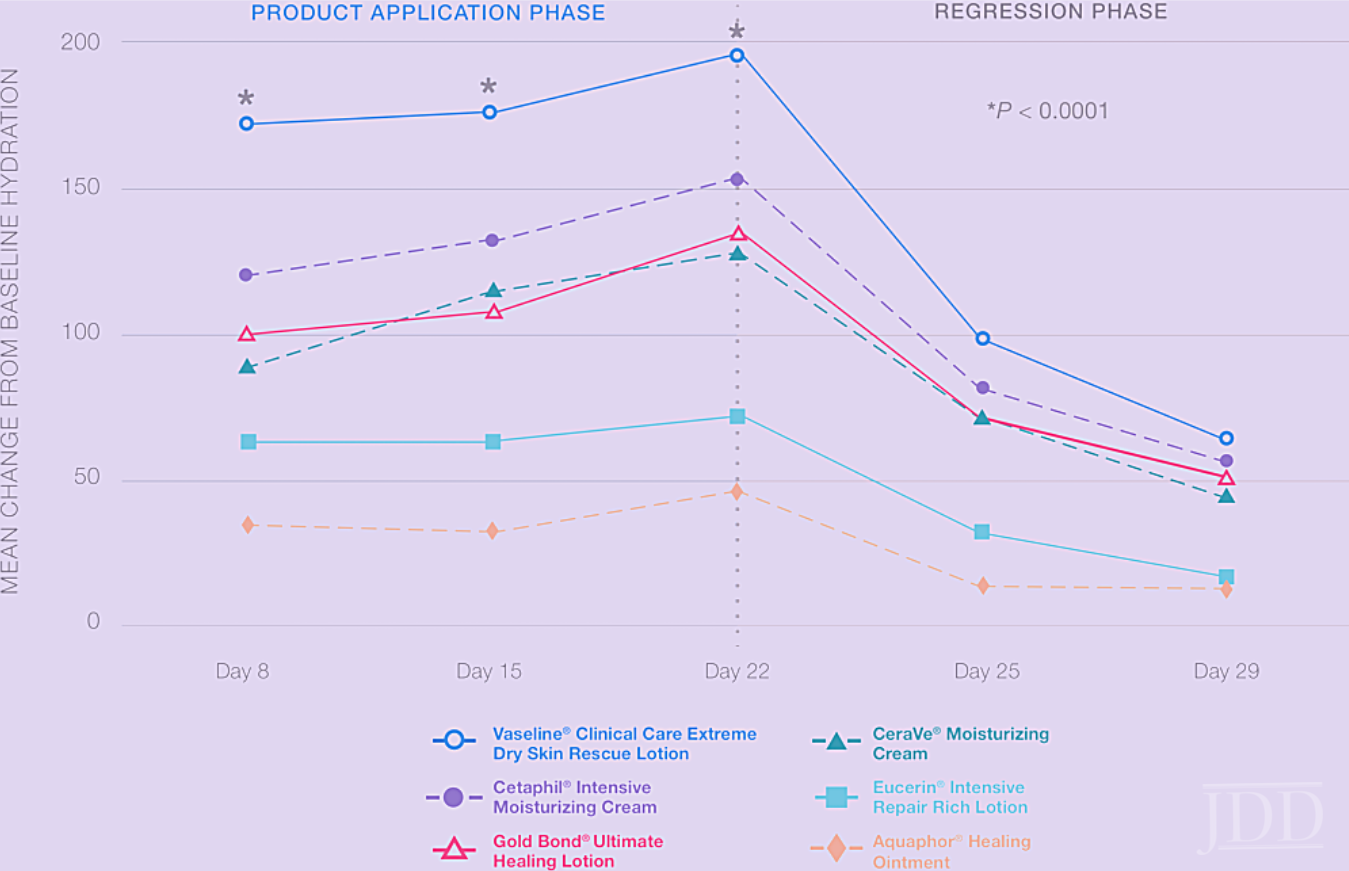
If there is no clear relationship between product format and efficacy, how does one select the right product? There are multiple considerations. First, it should be ensured that the product contains the right moisturizer combinations and levels (per INCI understanding as described earlier in part 1 of this series) to address the patient’s moisturization needs. Second, functional ingredients to address a patient’s secondary skin concerns should be considered and, based on personal experiences, specific ingredients that are not suitable for their skin or have caused issues or reactions in the past such as fragrances, should be avoided. Several key functional ingredients are described in more detail below. Finally, consumers should choose a product that meets their personal sensory preference and price tolerance so they can easily incorporate the product into their daily routine as using a product every day is a key factor to full realization of efficacy.
Functional Ingredients
In addition to the core skin lotion and cream ingredients described in part 1 of this series and the section above, it is common to include one or more bioactive ingredients that target specific pathways in the stratum corneum and underlying epidermis.
Niacinamide (Vitamin B3)
Niacinamide (Figure 8) is the physiologically active form of vitamin B3.This water-soluble vitamin has a variety of dermatological therapeutic benefits and is often included in many good moisturizers.
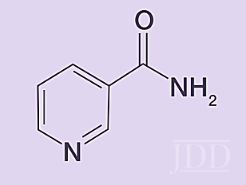
Niacinamide stimulates ceramide synthesis,13reduces hyperpigmentation,14,15provides anti-inflammatory and anti-bacterial benefits,16and contributes to anti-aging benefits like appearance of facial fine lines and wrinkles as proven at the concentrations used in the products (roughly 3%). One potential side effect of products containing niacinamide is flushing, particularly in consumers of Asian descent. The culprit ingredient is niacin, which is another form of vitamin B3 and can occur as a contaminant if the quality of the raw material is not properly controlled. Manufacturers of quality topical products know to screen raw materials for this contaminant.
Alpha Hydroxy Acid (AHA)
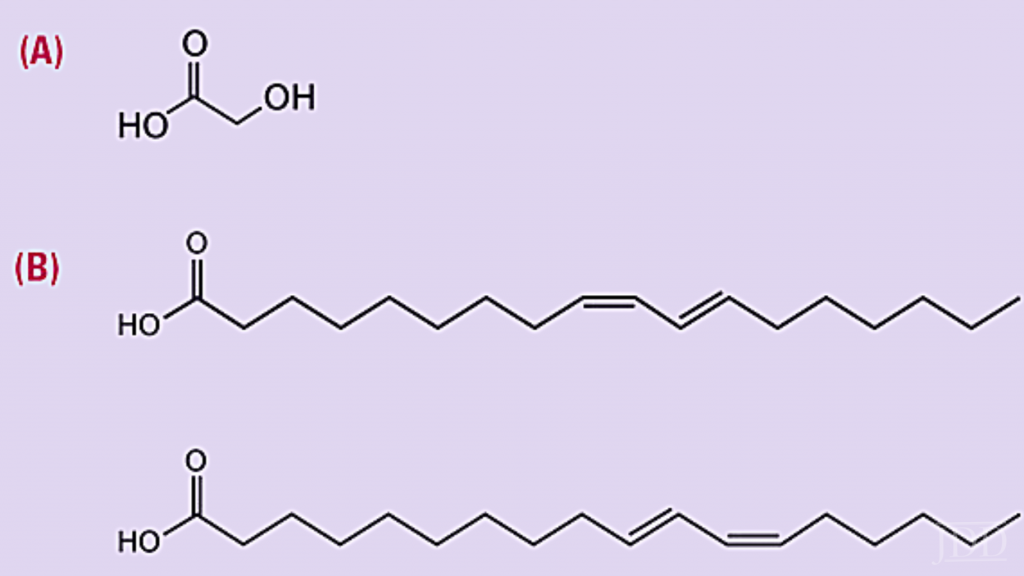
Alpha hydroxy acids (AHAs) are a class of organic carboxylic compounds including naturally-derived glycolic acid, lactic acid, malic acid, citric acid, and tartaric acid (Figure 9).17AHAs have been used at various concentrations to enhance desmosomes resolution and stimulate desquamation, with positive benefits for the epidermis and dermis.18,19,20At lower concentrations (5-10%), AHAs are available in over-the-counter products that can be used daily for improved barrier function and to improve the appearance of skin related to sun-damage, wrinkling, and hyperpigmentation benefits. At higher concentrations (20-70%), AHAs are used as chemical peels by dermatologists, beauty, and health spas.18
PPARs
Peroxisome proliferator-activated receptors (PPARs) are well recognized for their effects on skin barrier development and maintenance,21,22and on increasing keratinocyte differentiation.23,24,25They enhance the production of barrier important lipids such as ceramides and fatty acids,26,27and increase epidermal thickness and filaggrin synthesis leading to anti-aging benefits such as reduction in appearance of overall photodamage, mottled hyperpigmentation, and fine lines and wrinkles.28, 29
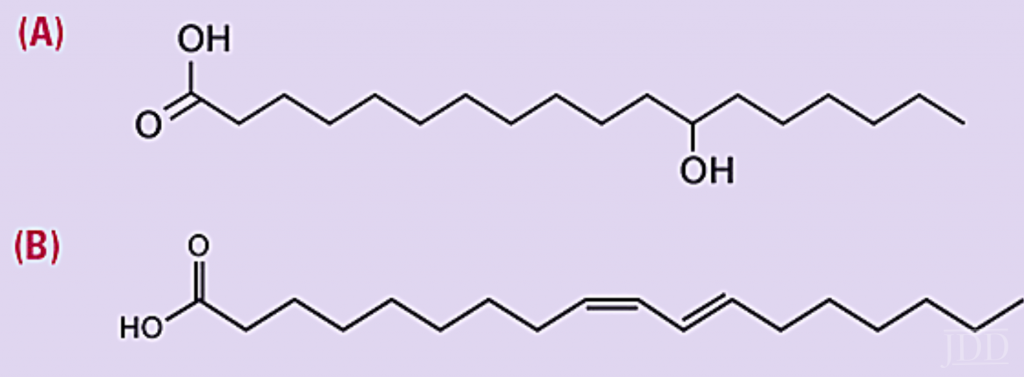
PPAR ligands are often naturally occurring unsaturated fatty ac- ids such as conjugated linoleic acid (CLA). Such compounds are unstable, rapidly oxidize when exposed to air, and lose ef- ficacy. Hence, historically, PPAR ligands have required the use of airless pack for stability leading to a higher cost product. In 2015, Unilever successfully introduced 12-hydroxy stearic acid (12-HSA) as a stable, gentle, and inexpensive PPAR ligand for the mass market (Figure 10). 12-HSA contains no unsaturated bonds obviating the need for high cost, airless packaging.
Vitamins C and E
The skin barrier is altered when exposed to external oxidative stresses such as UV irradiation and pollution.30Vitamin C (ascorbic acid) and E (α-tocopherol) are two of the most important antioxidants to protect skin from these external oxidative stresses. Although these are powerful ingredients, vitamins C and E are extremely unstable upon exposure to air. More stable forms of these vitamins like magnesium ascorbyl phosphate and vitamin E acetate are available; however, these need to be absorbed and converted into the active form within the skin. It is important to recognize that not all vitamin C and E formulas have similar physiological activities active.
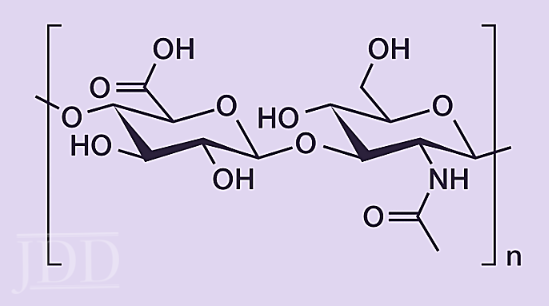
Hyaluronic Acid (HA), a disaccharide polymer (Figure 11), is an integral component of the extracellular matrix where it plays a key role in keratinocyte proliferation, migration, and wound repair. Because of the numerous -OH moieties, HA is exception- ally hygroscopic and is often touted as a wonder ingredient in skin-care products. However, due to its large molecular weight, topically dosed hyaluronic acid may not penetrate sufficiently into the skin for biological effects.
Stay tuned for part 3 of this series where product sensories and consumer experiences across moisturizing products will be discussed.
If you missed part 1 of this series, you can read it here.
References (click to display)
Source:
Lee, Christine, et al. “Principles of Moisturizer Product Design.” Journal of drugs in dermatology: JDD 18.1s (2019): s89-95.
Content and images used with permission from the Journal of Drugs in Dermatology.
Adapted from original article for length and style.
Did you enjoy this article? Find more on Derm Topics here.

