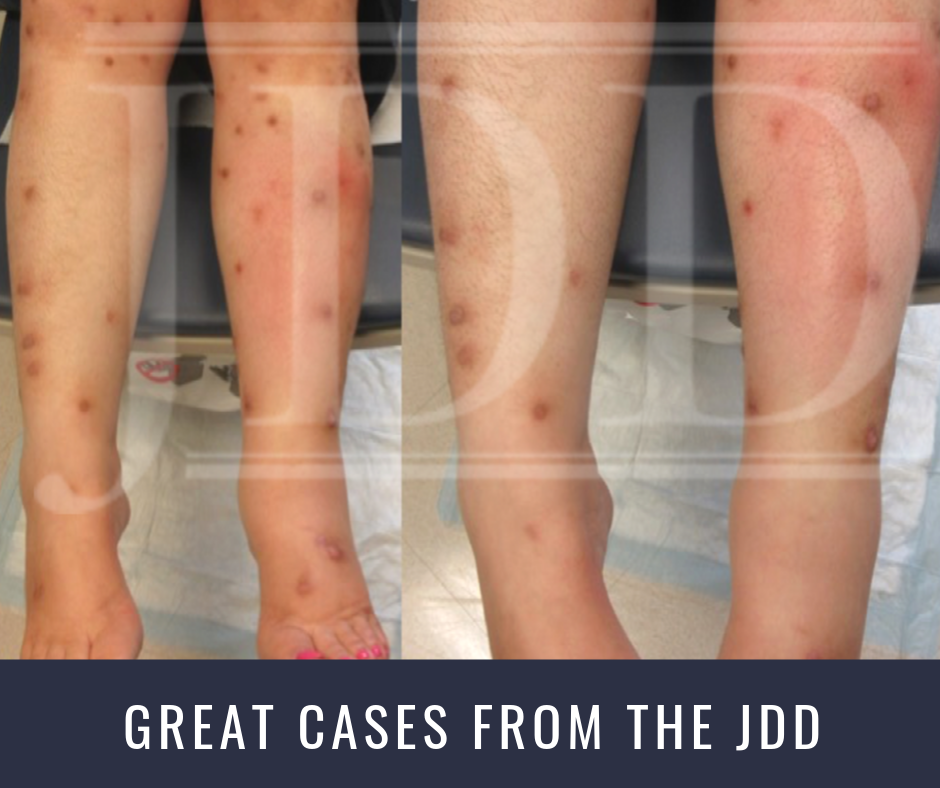
Authors Ross Tanis MD MS, Katalin Ferenczi MD, and B Michael Payette MD MBAB present a case where they demonstrated that treatment of Prurigo Nodularis (PN) with dupilumab significantly decreased pruritis and the size and number of new lesions after 2 months of treatment.
INTRODUCTION
CASE SYNOPSIS
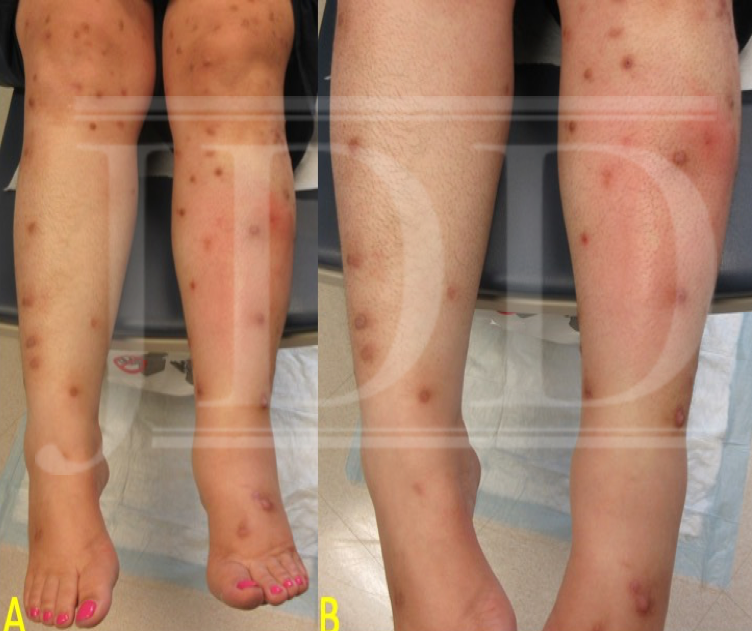
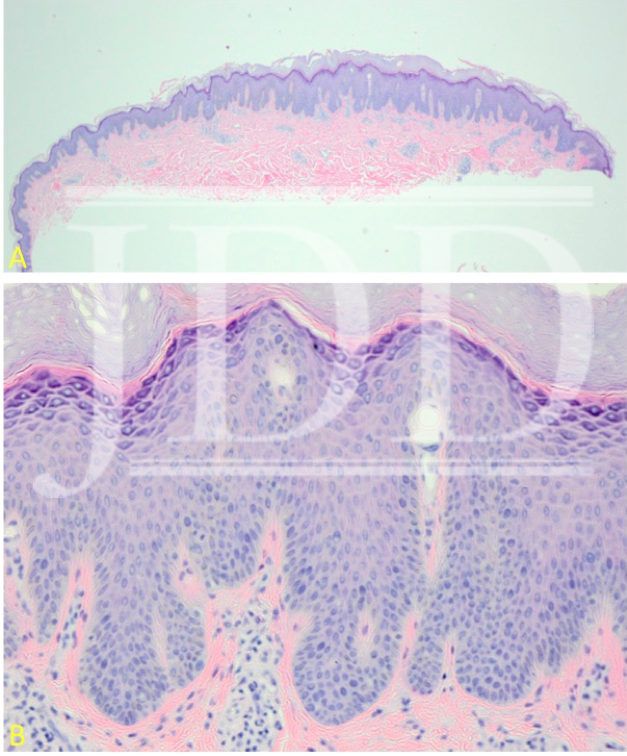
The patient was lost to follow-up for nearly 6 months, after which she returned with innumerable lesions. At that time, we elected to initiate dupilumab 600 mg subcutaneously (SQ) followed by 300 mg SQ every 2 weeks thereafter. At the 8-week follow up visit, the patient noted that some of the lesions had begun to diminish in size and become lighter in color and that new lesions were no longer developing. More importantly, she reported that her pruritis had significantly improved – by >50% – and she noticed she wasn’t scratching constantly. She noted that heat remained a trigger for itch. Finally, she noted that the return of her symptoms occurred most prominently 10 days after each injection. An attempt to increase to weekly dosing was made but was denied by the patient’s insurance.
CASE DISCUSSION
Prurigo nodularis most commonly affects middle-aged women, particularly African Americans.2 Individuals with a propensity for atopy and those with AD are more likely to develop PN, however, rarer causes of PN may be due to lichen planus, dermatitis herpetiformis, cutaneous T-cell lymphoma, among other entities.2 Atopic dermatitis is driven by the T-helper type 2 cell (Th2), in which interleukins (IL) -4 and -13 drive inflammation and pruritis and IL-31 is a potent pruritogen.3Indeed, lesional skin biopsies from patients with PN have shown a 50-fold increase in IL-31 mRNA levels compared to healthy controls.2
Individuals with atopy have increased innervation at lesional sites that may be due in part to impaired barrier function secondary to the itch-scratch cycle.4 Research by Oetjen et al. illustrated the role of the cytokines IL-4, IL-13 and IL-31 in the itch response. They demonstrated that IL-4 and IL-13 directly stimulate neurons that innervate the skin and that these same neurons also express IL-4Rα, IL-31Rα which serve a prominent role in the itch response. When IL-4 and IL-13 are intradermally injected they fail to elicit an itch response, unlike IL-31 which does elicit an itch response when injected both intradermally and intrathecally. However, when the IL-4Rα was deleted on sensory neurons, there was a marked reduction in the itch response, suggesting that IL-4 and IL-13 do not directly induce an itch response but rather sensitize the neurons to the itch response produced by pruritogens such as IL-31.4
Dupilumab is a fully human monoclonal antibody (mAB) that blocks (receptor antagonist) the IL-4Rα and subsequent IL-4 and IL-13 signaling.5 In a randomized, placebo-controlled clinical trial in patients with moderate-to-severe AD, dupilumab resulted in drastic improvement of patient-reported pruritis using the pruritis numerical rating scale and the 5-dimension pruritis scale.5 To demonstrate the role of IL-31 in pruritis, nemolizumab, an anti-IL-31 mAB, was shown to reduce itch in patients with moderate-to-severe AD using the visual analogue scale in a randomized, double-blind, placebo-controlled trial.6
Due to disease overlap and similar pathophysiologic mechanisms related to itch between PN and AD, it was reasonable to hypothesize that dupilumab treatment would benefit our patient. After 8 weeks of therapy with dupilumab, the patient reported that she was developing fewer lesions and current lesions had begun to reduce in size. Moreover, she had significant improvement in her pruritis, albeit not symptom free. Interestingly, she indicated that the pruritis increased in intensity near the end of the biweekly dosing schedule, which prompted us to increase the dosing to weekly, but as mentioned, this was denied by the patient’s insurance. Nonetheless, this illustrates an important concept in that when a successful treatment option has been uncovered for an off-label indication, adjusting the dose and/or frequency of the medication may be beneficial. It’s quite plausible that varying doses and/or frequency of dupilumab will be optimal for different diseases (i.e. adalimumab dosing is different for psoriasis vs hidradenitis). Due to the inability to obtain weekly doing in this case, the potential for added benefit here remains unknown.
The versatility of dupilumab has been illustrated with the treatment of multiple other dermatologic conditions. Case reports of alopecia areata,7 bullous pemphigoid,8 eosinophilic annular erythema (EAE),9 and dyshidrosis10 have been shown to successfully respond to dupilumab treatment – as early as 1 month in EAE vs 9 months in alopecia areata. There are also sparse cases of dupilumab for prurigo nodularis.11,12,13 Central to the role of dupilumab in the treatment of these disorders is the commonality of Th2 immunity in the pathogenesis. The blockade of the IL-4Rα receptor by dupilumab illustrates the importance of this receptor in Th2 cell activation and its role in the pathogenesis of many diseases. Furthermore, this demonstrates that dupilumab is a logically viable treatment option for patients with recalcitrant pruritic diseases that are driven by the pathogenesis of Th2 cells.
CONCLUSION
Treatment of recalcitrant prurigo nodularis with dupilumab reduces the severity of the pruritic symptoms and the quality and number of papulonodules. Dupilumab should be considered as a viable treatment option in prurigo nodularis when standard therapy regimens have failed after and adequate trial. Additionally, the use of dupilumab has been demonstrated in other cutaneous disorders where the pathophysiology is mediated by a Th2 helper cell response. Future investigations into the redundant role of the IL-4Rα and the dampening of the Th2 helper cell response will allow dupilumab to gain expanded FDA approval for these recalcitrant cutaneous disorders.
References: click to display.
Source:
Tanis, R., MD MS, Ferenczi, K., MD, Payette, M.,MD MBAB (2019). Dupilumab Treatment for Prurigo Nodularis and Pruritis. Journal of Drugs in Dermatology, 18(9), 940-941. https://jddonline.com/articles/dermatology/S1545961619P0940X/1/
Content and images used with permission from the Journal of Drugs in Dermatology.
Adapted from original article for length and style.
Did you enjoy this case report? Find more here.