Introduction
Dermoscopy, also known as epiluminescence microscopy, epiluminoscopy or skin surface microscopy, is a noninvasive technique for examination of skin by using a high quality magnifying lens and powerful lighting system to visualize the skin (Figure 1). Although dermoscopy was initially used for the study of mainly pigmented lesions, in the past several years its utility in non-pigmented skin tumors, inflammatory conditions and certain skin infections has become evident. Part one of this article will focus on dermoscopy with evaluation of pigmented lesions. Part two will focus on vasculature, specific features in non-pigmented skin tumors, and both inflammatory and infectious diseases.

Polarized Light and Immersion Fluid
Many dermatoscopes allow for the option of using non-polarized light and polarized light along with contact and non-contact dermoscopy. Polarized dermoscopy (PD) allows for better visualization of deeper structures (located at the dermoepidermal junction [DEJ] or superficial dermis) such as vessels and collagen and is essentially ‘blind’ to superficial layers, while non-polarized light is better at visualizing superficial layers of the skin, such as comedo-like openings in seborrheic keratosis. If using contact dermoscopy, use of immersion fluid such as 70% isopropyl alcohol can enhance the image by reducing light reflection from the skin surface.
Colors (Figure 3)
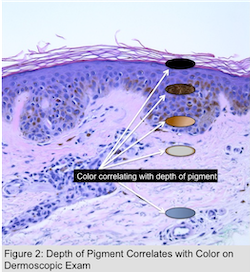
An important aspect of dermoscopy is the significance of different colors seen with dermoscopy, as the depth of pigment often determines color visualized on exam (Figure 2). For example, the darkest color (black) corresponds to superficial pigment in the upper layers of the epidermis (stratum corneum, stratum spinosum), while a medium brown typically occurs when melanin is found at the DEJ or lower epidermal layers. When the pigment is deeper (papillary dermis) the color is often slate blue (i.e. pigment drop in lichen planus), while the deepest location (reticular dermis) conveys a steel blue color (blue nevus).

Pigmented Lesions
Dermoscopy is perhaps the most helpful in pigmented lesions, which can be melanocytic or non-melanocytic in origin. To help determine if melanocytic in origin, the following features are helpful as melanocytic lesions typically have the presence of one of the following: reticular pigment network, dots or globules, structureless pigmentation, or other location/lesion specific pattern.
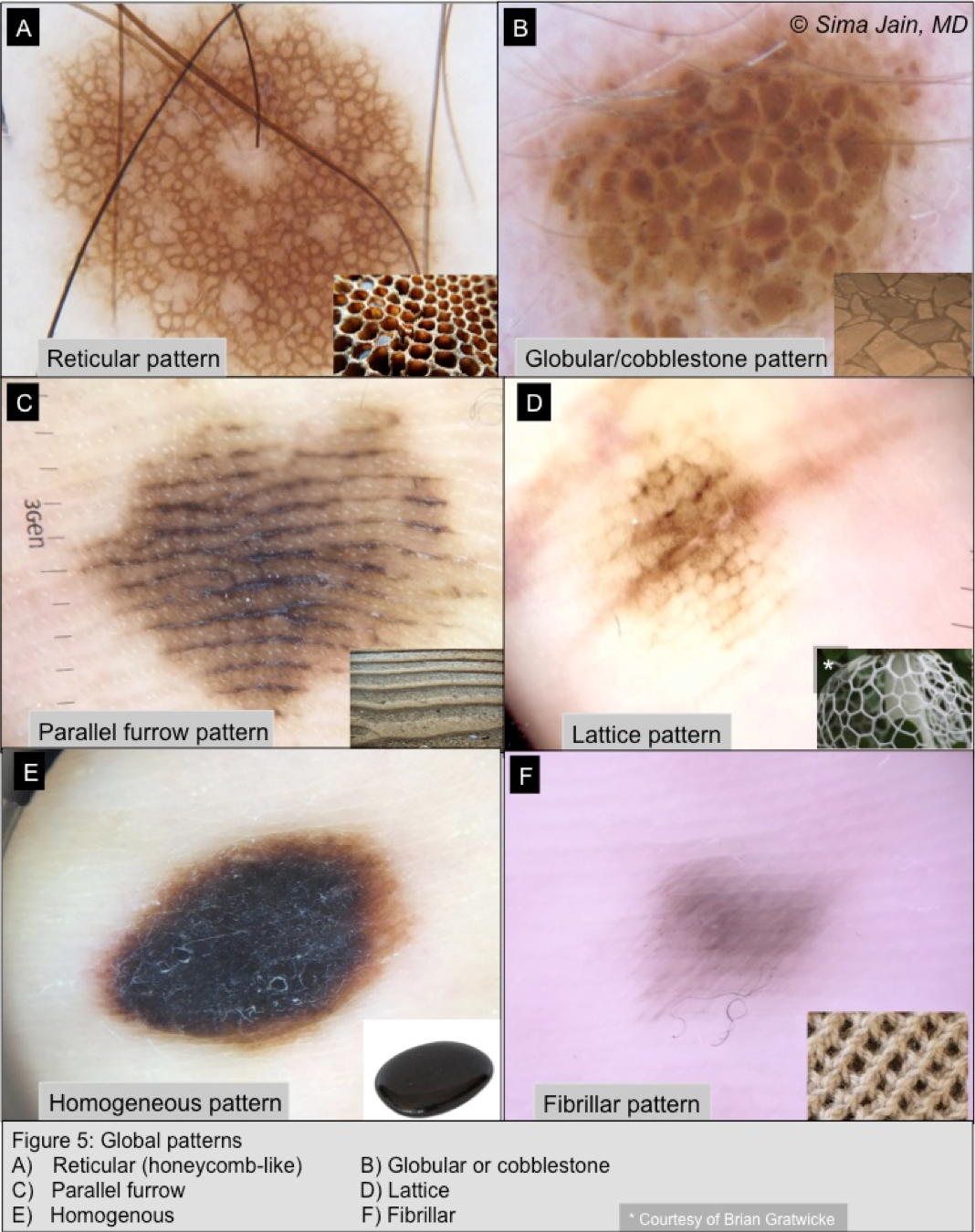
Global Patterns of melanocytic lesions (Figure 5): 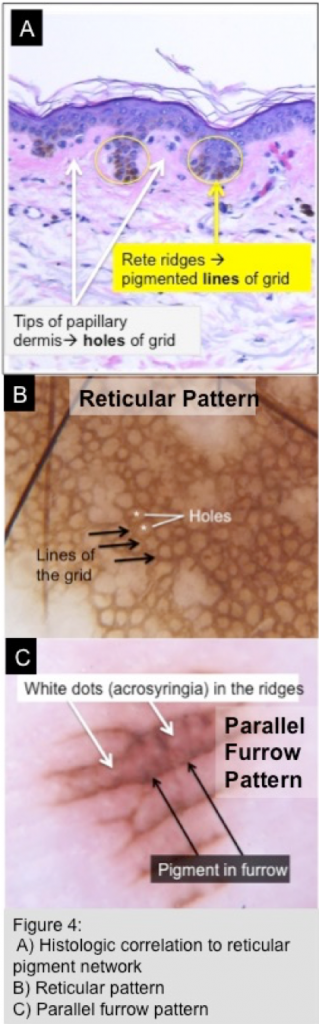
-
- Reticular pigment network (Figures 4B, 5A)
- Most common network (typical) seen in nevi and consists of a honeycomb-like pattern with dense pigment rings resembling a grid; the ‘holes’ correspond to the tips of the papillary dermis and the pigment correlates with the rete ridges containing increased melanin (Figure 4A)
- Benign nevi have a symmetric grid that is evenly spaced out with gradual fading of the network at the periphery (Figure 5A)
- Dots and globules (Figure 5B)
- Regular dots or globules regularly distributed throughout nevus
- Homogenous pattern (Figure 5E)
- Diffuse brown, blue or gray-blue pigmentation of lesion without overt pigment network or other overt structures; due to homogenous distribution of melanocytes (can be seen when rete ridges flatten out or with bridging of pigment-laden ridges)
- Location/lesion specific:
- Parallel furrow pattern (Figures 5C): pigment seen in the furrows (sulci) but not seen in the ridges; pattern commonly seen in acral sites; of note, non-pigmented dots (acrosyringia) seen in ridges so pigment should spare this area (see Figure 4C)
- Lattice pattern (Figure 5D): parallel pigmented lines along furrows with lines crossing the ridges in a lattice pattern; variant of parallel furrow and also found in acral sites
- Fibrillar pattern (Figure 5F): filamentous pattern with mesh-like arranged fibrillar pigmentation due to pigmented lines crossing skin markings diagonally; often found in acral sites
- Starburst pattern: radial uniform streaks at periphery of lesion
- Multicomponent global pattern: consists of combination of pigmentation pattern; often seen in melanoma
- Reticular pigment network (Figures 4B, 5A)
Pseudonetwork (Figure 6)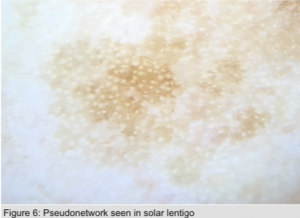
The term pseudonetwork is used to define homogenous pigmentation interrupted by nonpigmented ostia of adnexal structures characteristically seen on the face. It is mainly found in nonmelanocytic pigmented lesions on the face such as a solar lentigo, but it can also be seen in melanocytic lesions as well. It is not a true pigment network as it is essentially junctional pigment that forms a broad mesh with white holes created by terminal/vellus follicles and sweat gland openings.
Negative Pigment Network (Figure 7)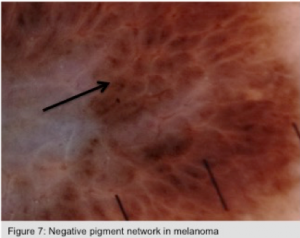
The term negative pigment network, also known as inverse network, reverse network or reticular depigmentation, reveals darker ‘holes’ with lighter areas filling the grid of the network. Lighter grid lines appear serpiginous but darker areas resemble elongated curved or tubular globules. This particular network is highly specific for melanoma, especially when focal and asymmetric, but it can also be seen in dermatofibromas, Spitz nevi and rarely in other nevi (i.e. congenital).
Benign vs. Malignant Pigmented Lesions (Figure 8)
For evaluating dermoscopic images of pigmented lesions, there are four main diagnosis methods: pattern analysis, ABCD rule, Menzies method and 7-point checklist. We will go over pattern analysis here as this is considered the classic approach to diagnose melanocytic lesions and to differentiate between benign and malignant tumors. Pattern analysis is the overall assessment of the diagnostic value of features seen by dermoscopy.
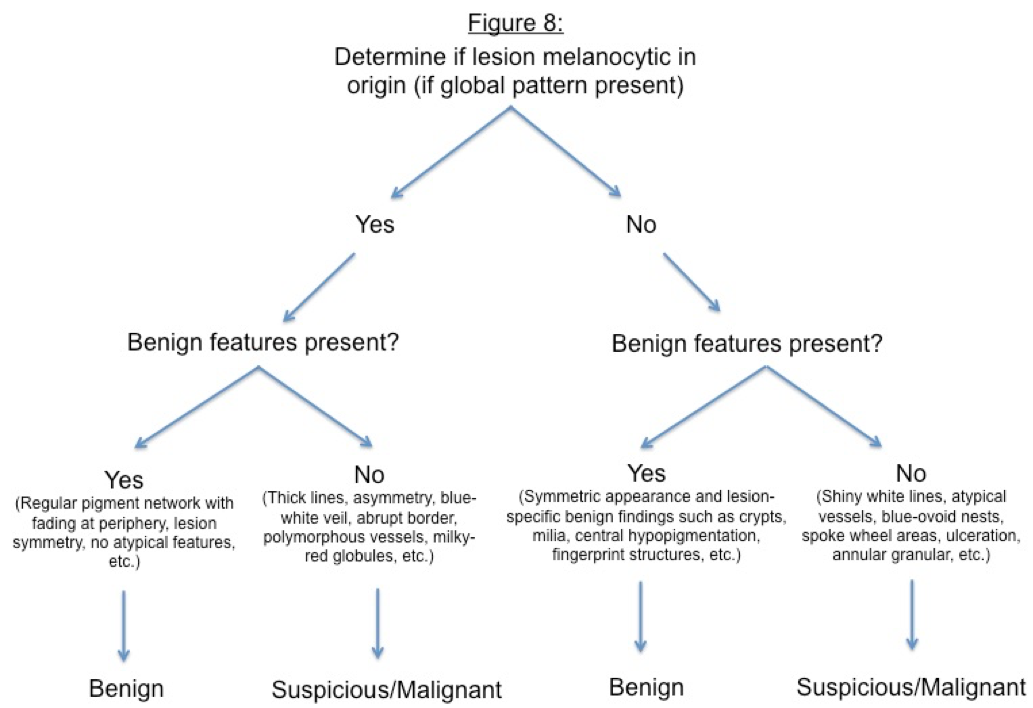
- Step 1: Look at the global pattern of the lesion to determine if origin melanocytic
- If able to identify global pattern (reticular, dots/globules, homogenous, etc.) → melanocytic in origin go to → next step to identify if benign or suspicious/malignant
- If not able to identify global pattern → likely not melanocytic in origin and pigment may be related to excess melanin in lesion (i.e. seborrheic keratosis, solar lentigo, pigmented basal cell carcinoma (BCC), pigmented AK, dermatofibroma, pigment drop in lichen planus/keratosis, etc.) → proceed to second step to find characteristic features to help confirm specific diagnosis
- Step 2: Look at the local features of the lesion
- If melanocytic:
- Evaluate the pigment network, for any characteristic features and the overall appearance
- Pigment network: is it symmetric or asymmetric, one component or multicomponent, typical or atypical network, fades at periphery or sharply cut off
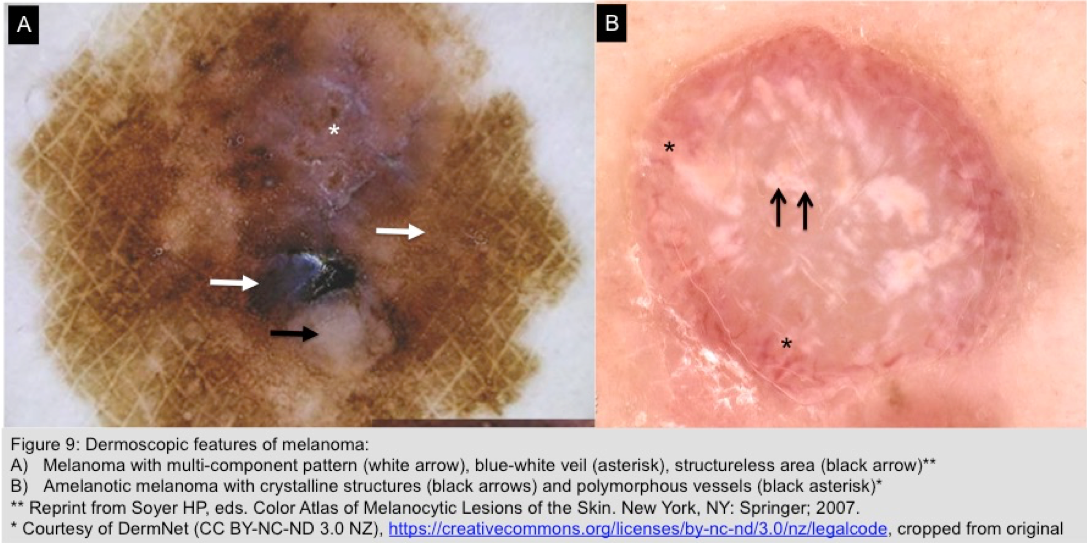
- Look for atypical dermoscopic features associated with melanoma (Figure 9):
- Atypical pigment
- Branched streaks
- Radial streaming
- Pseudopods
- Thick lines
- Sharp cut off of network at periphery
- Irregular dots/globules/blotches
- Blue-white veil
- Other atypical features
- Crystalline structures (shiny bright white lines)
- Milky-red areas
- White structureless areas (regression)
- Polymorphous vessels
- Atypical pigment
- Evaluate the pigment network, for any characteristic features and the overall appearance
- If melanocytic:
-
-
- If non-melanocytic, look for specific features to help identify, for example:
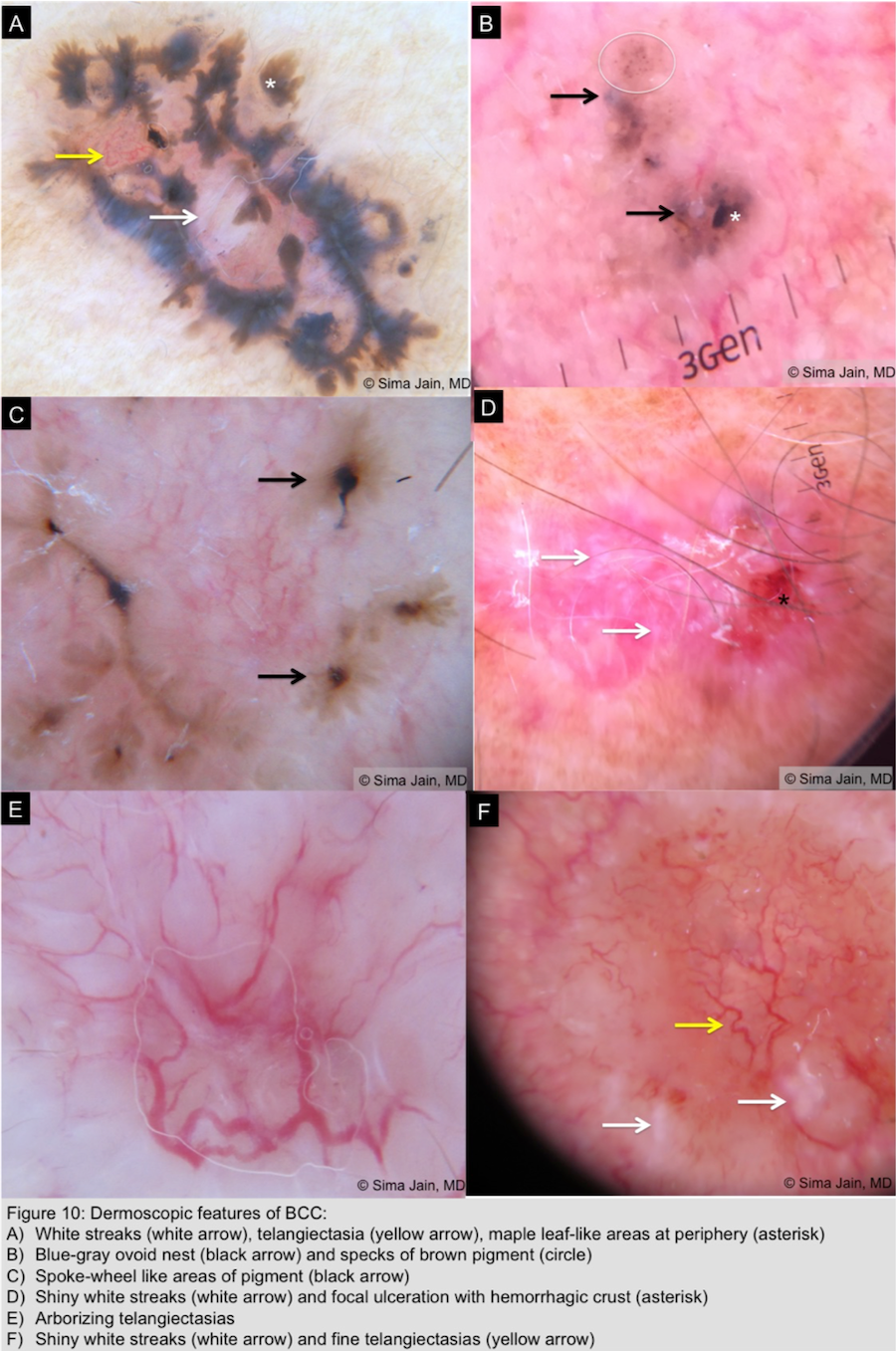
- Pigmented BCC (Figure 10)
- Absence of pigment network
- Blue-gray ovoid nests or blotches
- Specks of brown and grey pigment
- Spoke wheel-like areas of pigment (central hub with radial projections)
- Leaf-like areas on the periphery of the lesion
- Hypopigmented and red structureless areas
- Crystalline structures (shiny bright white lines)
- Focal hemorrhage-ulceration
- Arborizing and atypical vessels
- Pigmented BCC (Figure 10)
- If non-melanocytic, look for specific features to help identify, for example:
-
-
-
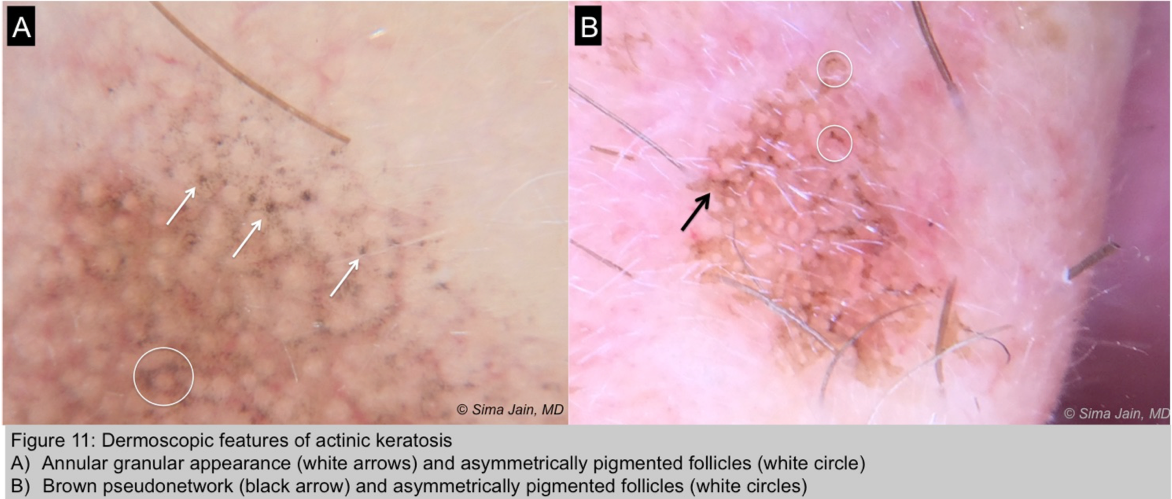
- Pigmented actinic keratosis (Figure 11)
- Multiple gray to dark brown dots around follicular ostia
- Rhomboidal structure: thickened gray to dark brown areas around follicular ostia in shape of rhomboid
- Annular-granular structure: multiple coarse dots around ostia with an annular granular appearance
- Pseudonetwork: confluence of annular-granular and rhomboidal structures resulting in a gray or brown pseudonetwork
- Asymmetric pigmented follicles: asymmetric gray to brown rings of pigmentation
- Multiple gray to dark brown dots around follicular ostia
- Pigmented actinic keratosis (Figure 11)
-
-
-
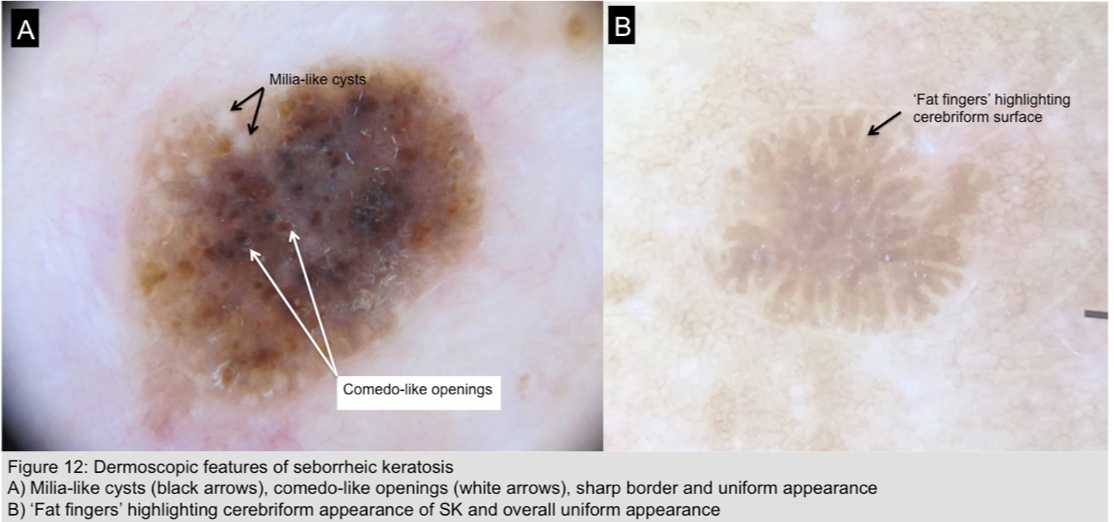
- Seborrheic keratosis (Figure 12)
- Milia-like cysts and comedo-like openings
- Cerebriform surface with ‘fat fingers’ – thick linear/curvilinear/branched structures representing gyri of the cerebriform surface
- Sharp demarcation ± moth-eaten border
- Seborrheic keratosis (Figure 12)
-
-
-
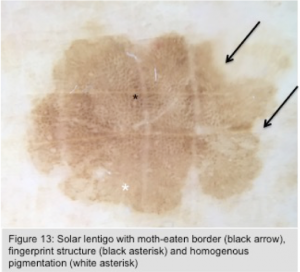
- Solar lentigo (Figure 13)
- Homogenous light brown pigmentation ± faint area of reticulation
- Sharp demarcation with moth-eaten border
- Fingerprint-like areas
- Pseudonetwork
- Solar lentigo (Figure 13)
-
-
-
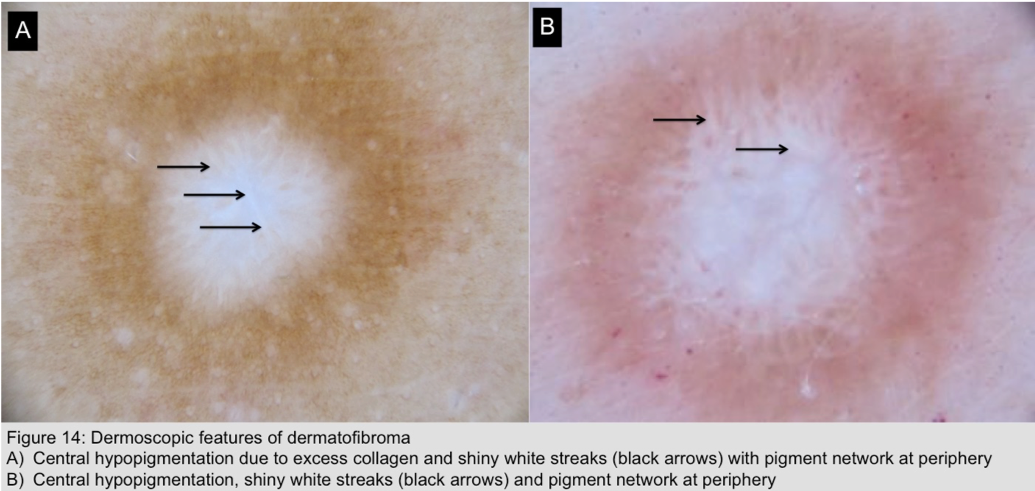
- Dermatofibroma (Figure 14)
- Central white area surrounded by faint network or pseudonetwork
- Crystalline structures (shiny white linear streaks due to excessive collagen)
- Dermatofibroma (Figure 14)
-
Part two of this 2-part dermoscopy review will focus on the importance of vasculature and the specific morphological features and patterns that help guide accurate diagnosis. It will also cover characteristic findings for specific conditions, including but not limited to verruca vulgaris, molluscum contagiosum, scabies, and psoriasis. Go to Part Two.
Did you know? Dr. Sima Jain will be leading a Dermoscopy workshop at the upcoming ODAC Dermatology, Aesthetic, Surgical Conference(ODAC) on Saturday, January 20th at the JW Marriott Grande Lakes in Orlando, FL. To learn more and register, click here. ODAC and Next Steps in Derm are products of SanovaWorks.
References:
-
-
- Lallas A, Giacomel J, Argenziano G, et al. Dermoscopy in general dermatology: practical tips for the clinician. Br J Dermatol. 2014 Mar; 170(3): 514-26.
- Soyer H, Argenziano G, Zalaudek I, et al. Dermoscopy: the essentials. Philadelphia, PA: Saunders Ltd, 2012.
- Paech V, Schulz H, Argenyi Z, et al. Compendium of Surface Microscopic and Dermoscopic Features. Heidelberg, Germany: Springer-Verlag, 2008.
- Russo T, Piccolo V, Ferrara G, et al. Dermoscopy pathology correlation in melanoma. J Dermatol. 2017 May;44(5): 507-514.
- Neila J, Soyer HP. Key points in dermoscopy for diagnosis of melanomas including difficult to diagnose melanomas on the trunk and extremities. J Dermatol. 2011 Jan;38(1): 3-9.
- Liebman T, Rabinovitz H, Balagula Y, et al. White Shiny Structures in Melanoma and BCC. Arch Dermatol. 2012; 148(1): 146.
- Menzies SW, Moloney FJ, Byth K, et al. Dermoscopic Evaluation of Nodular Melanoma. JAMA Dermatol. 2013;149(6):699–709.
- Balagula Y, Braun RP, Rabinovitz HS, et al. The significance of crystalline/chrysalis structures in the diagnosis of melanocytic and nonmelanocytic lesions. J Am Acad Dermatol.2012 Aug; 67(2):194.
- Salerni G, Alonso C, Fernandez-Bussy, Ramon. Crystalline Structures as the Only Dermoscopic Clue for the Diagnosis of Basal Cell Carcinoma. Arch Derm. 2012 Jun; 148(6): 776.
- Kelati A, Baybay H, Moscarella E, et al. Dermoscopy of Pigmented Actinic Keratosis of the Face: A Study of 232 Cases. Actas Dermosifiliogr. 2017 Nov; 108(9): 844-851.
-