Dr. Friedman, jack of many trades, Professor and Interim Chair of Dermatology at George Washington University provided an excellent overview of ways to achieve victory over atopic dermatitis (AD). This expert discussion took place during the ODAC Inflammatory Diseases Symposium on December 3, 2020.
How could we possibly cover atopic dermatitis in 20 minutes? Well, Dr. Friedman skillfully led us through epidemiology, education, and treatment of this disease, including the “Easy Es” or really 4 “Es” of atopic dermatitis management:
1. Education: Soaps, baths, and moisturizers, oh my!
-
-
- Hint: less is more, use soaps/cleansers with neutral or acidic pHs (closer to our natural skin pH for optimal function of enzymes in our skin)
- Avoid soaps entirely during flares!
- Moisturizers: more is better!
-

2. Expectations: there is no cure!
-
-
- We must treat during different phases of therapy: ie go “hard” (high potency) when you need to1!
-

3. Encouragement: you can do it! (but let’s follow up in a few weeks to check-in)
-
-
- Proactive therapies with nonsteroidal options are very important. Treat normal skin to prevent relapse.
-
Current topical therapies VS. Upcoming topical therapies


4. Enough of an effective medication: aka, how do we address the patient that has “failed everything” or “nothing works!”
-
-
- Friedman gave us several clinical pearls on how to promote patient compliance, including use of topicals in maintenance vs “flare” periods
- Once-daily application of mid to high potency agents may be as effective as twice daily (yet at a lower cost)
-
Other treatment pearls:
-
- For acute flares: apply topical corticosteroids to the inflamed skin 3 7 days beyond clearance
- If acute flare is not resolved in 7 days, consider nonadherence, infection, misdiagnosis, or contact allergy
- Don’t be afraid to perform a KOH prep and/or biopsy untreated skin (at least 3 weeks off treatment)
- If still flaring despite accounting for the above factors, consider referral to an atopic dermatitis specialist and/or step-up treatment
- Provide written instructions/ an “eczema action plan”
- Reassess control every 4-8 weeks for the first 3 months
On to the details of Dr. Friedman’s lecture:
1. Facts about atopic dermatitis2-5:
31.6 million people in the United States have some form of eczema
25-35% of cases persist from childhood to adolescence or adulthood
Eczema causes worse impairment in quality of life than autoimmune disorders, diabetes, food allergy, and heart disease
Patients with atopic dermatitis experience PAIN
Remember to document quality of life impairment and pain in your clinic notes. Specifically, ask:

2. What’s the deal with diagnosis?
In 2014, the AAD released a 4-part series of guidelines6-9 for atopic dermatitis, which included diagnostic criteria (see below). Remember, atopic dermatitis is a clinical diagnosis.
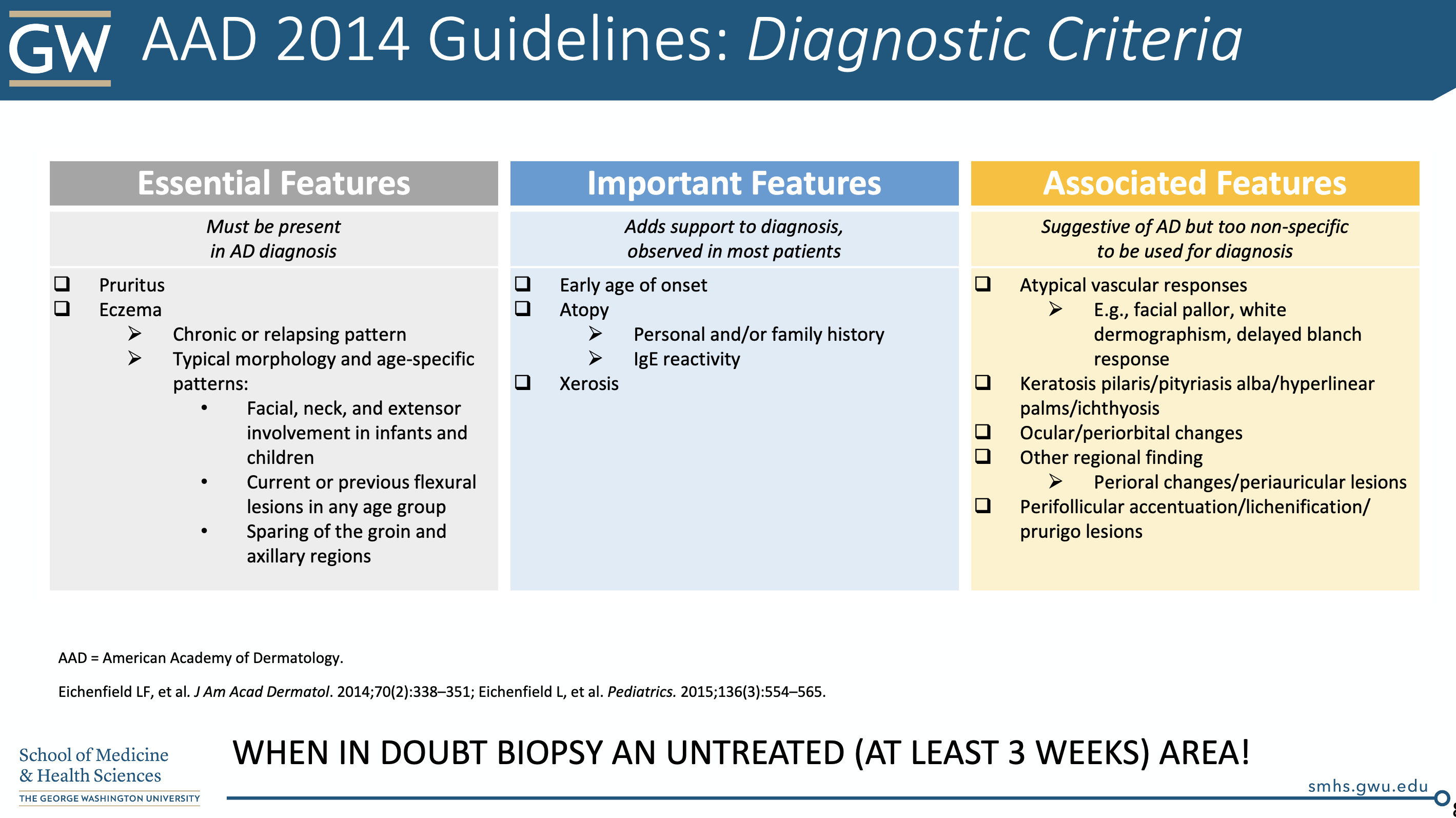
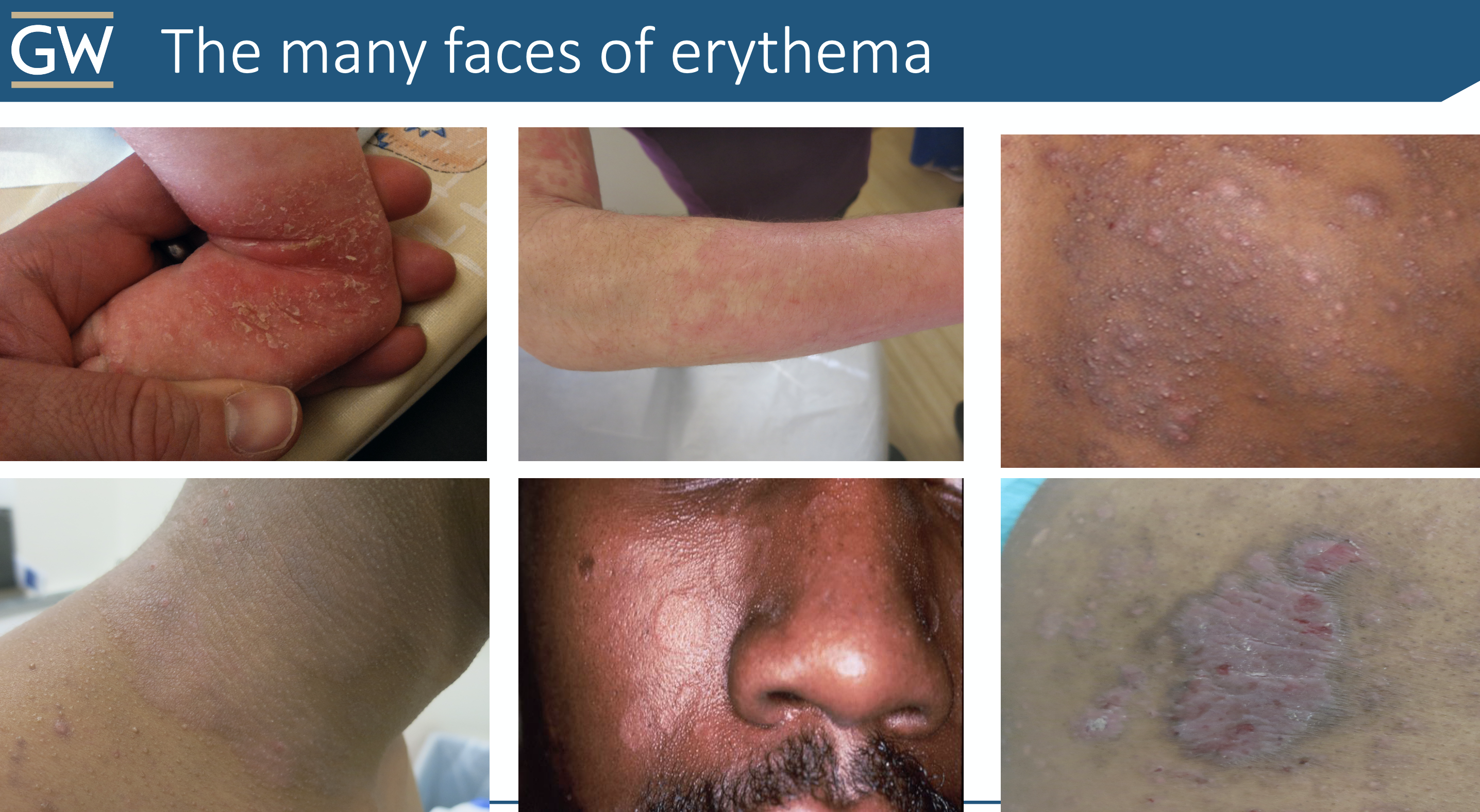
Don’t be fooled by erythema in different skin types (spoiler alert: there is a pop quiz at the end!)
3. Basic Principles of Treatment
Use a stepwise approach to managing atopic dermatitis10. We need both an active and long-term treatment plan.
Non-lesional skin: focus on gentle skincare and trigger avoidance
Mild disease: add in antiseptic measures
Moderate disease: add in topical corticosteroids or steroid-sparing agents
Severe disease: consider adding systemic therapy or phototherapy, referral to an atopic dermatitis specialist, short term hospitalization

4. Educating our patients:
1. Soaps: pH matters! “True soaps” tend to have a higher (more basic) pH and act as surfactants (stripping the natural oils of the skin).
High pH activates proteases (break down the barrier of the skin), increases Strep pyogenesand Staph aureus colonization, and removes important cholesterol-based molecules from the skin.
Instead, use neutral to low-pH moisturizing cleansers, soap-free cleansers, or modified surfactant cleansers.
Examples:
-
- Low pH cleanser: Dove (pH =7), Cetaphil (soap free, pH=6)

- Low pH cleanser: Dove (pH =7), Cetaphil (soap free, pH=6)
-
- Modified surfactant: Aveeno

2. Bathing
-
- Do not rub, scrub, or use washcloths (this tends to blow patient’s minds)
- Limit cleanser use (No fragrance! Hypoallergenic! No additives!)
- There is no standard for frequency, type, or duration of bathing in patients with atopic dermatitis
3. Moisturizers
-
- Moisturize, moisturize, moisturize. Try to put on the moisturizer within 30 minutes of bathing.
Take a look at Dr. Friedman’s slide below regarding the difference between emollients vs occlusive vs humectants (he wants all 3 for his AD patients!)

Just in case we didn’t believe enough in moisturizers, Dr. Friedman supplied us with two different RCTs supporting the use of emollients in infants at high risk for AD.
The first study, a 2014 article published in the Journal of Allergy and Clinical Immunology, was a RCT performed on 124 neonates from the US and UK who were high risk for atopic dermatitis (eg. parent(s) with eczema, any filaggrin mutation)11. The intervention arm was randomized to apply emollients at least once per day starting within 3 weeks of birth. The control arm was instructed not to use emollients. Participants were followed to determine incidence of atopic dermatitis by 6 months. The intervention group had a relative risk reduction of 50% (95% CI, 0.28-0.9, p=0.017) in incidence of AD.
The second study, based out of Japan, was a RCT of neonates in which 59/118 neonates at high risk of atopic dermatitis applied an emulsion-type moisturizer daily for the first 32 weeks of life12. At 32 weeks, 32% fewer infants in the intervention group had atopic dermatitis (p=0.012).
What about Vaseline? (100% white petrolatum)
Plain petrolatum increases filaggrin expression, increases antimicrobial peptides, AND reduces T cell inflammation.
A 2016 study was published in the Journal of Allergy and Clinical  Immunology13 involving two cohorts applying 100% white petrolatum. The study analyzed expression of innate immune genes, antimicrobial peptides, cytokines, skin thickness, filaggrin and loricrin (among others) in patient skin samples on which petrolatum was applied.
Immunology13 involving two cohorts applying 100% white petrolatum. The study analyzed expression of innate immune genes, antimicrobial peptides, cytokines, skin thickness, filaggrin and loricrin (among others) in patient skin samples on which petrolatum was applied.
The first cohort of patients were 13 patients with AD and 16 patients without AD. Nonlesional/unaffected skin in both groups was occluded with 100% white petrolatum using a Finn chamber (paper discs commonly used to apply contact allergens in patch testing). The occlusive Vaseline was placed for 48hrs, and two biopsies were taken at the 72hour mark – 1 of occluded skin + Vaseline, 1 of nonoccluded/nonlesional skin). A second cohort of healthy patients was enrolled to further compare the time points of maximal gene induction, with multiple biopsies comparing control skin vs skin occluded with Finn chamber only vs skin occluded with Vaseline+Finn chamber at 48 and 72 hours.
Occlusion of non-lesional skin with 100% petrolatum resulted in:
-
- upregulation of innate immune genes
- increased expression of antimicrobial peptides
- increased expression of filaggrin and loricrin
- increased stratum corneum thickness
- reduced T-cell infiltrates
What about Aveeno (colloidal oatmeal)?
It is a “prebiotic” for our skin flora! (and so much more…see below)
Did you know that oat supports the growth of commensal microorganisms, such as S. epidermidis, but NOT S. aureus14? Colloidal oatmeal also has anti-inflammatory, anti-pruritic, anti-oxidant, anti-fungal, and barrier repair properties as well as beneficial effects on skin pH (increased production of lactic acid)14. Oat is rich in phytochemicals (including ferulic acid and caffeic acid), as well as sugars, amino acids, proteins, lipids, and fiber, which act as “nourishment” for our native skin flora.
Emollients can also offer oligoelements such as calcium, magnesium, manganese, strontium, and selenium.
A study published in 2014 in the Journal of Drugs in Dermatology demonstrated that an emollient containing shea butter, niacinamide, and LaRoche-Posay thermal spring water significantly improved diversity of skin microbiota in lesional skin of patients with atopic dermatitis15.
5. Topical therapy for atopic dermatitis: the 411
-
- Topical corticosteroids
The goal of topical corticosteroid therapy is to treat flares and then maintain the skin to reduce the frequency of flares.
Let’s start with some of our typical topical steroid regimens:
A standard regimen can consist of:
-
- “Hit ‘em strong”
Once to twice daily application of topical steroids for 2-3 weeks, then PRN thereafter. The local side effects (ie atrophy, telangiectasias) actually come with duration of use more-so than potency of steroid, so don’t be afraid to use high potency corticosteroids.
Note: once-daily application of mid-to-high potency topical steroids may be just as effective as twice-daily application16. Save your patient’s money while improving compliance!
-
- Then “Maintain”
After the flare is controlled, a maintenance regimen can be established. This often consists of applying a corticosteroid 2-3 times a week or incorporating steroid-sparing agents.
If disease is severe:
-Consider adding pulse doses of super potent steroids on the weekends
In the most severe cases:
-Incorporate a super potent steroid 3 times a week during the maintenance phase
2. Topical calcineurin inhibitors. See Dr. Friedman’s slide below for the details.

3. Crisaborole (EUCRISA®)
Crisaborole is a topical phosphodiesterase 4 inhibitor, which acts to increase intracellular cAMP, which is associated with reduced inflammation. Further, this topical has a low molecular weight so it can better penetrate the skin barrier.
NOTE: patients can feel a burning sensation with both topical calcineurin inhibitors as well as crisaborole. We can counsel our patients that this is typically temporary and intensity is often site-specific. Make sure to prepare your patient for this potential side effect.
4. The new kids on the block: JAKs and more
The Janus Kinase signaling system mediates the biological effects of several key cytokines implicated in atopic dermatitis, including IL-4, IL-13, and IL-31.
Specifically, janus kinase is a family of intracellular, non-receptor tyrosine kinases. The family consists of JAK1, JAK 2, JAK 3, and TYK2—tyrosine kinase 217. There are different oral and topical medications that inhibit the JAKs to different degrees.
For example, tofacitinib (oral, and now compounded into topical formulations, approved for RA, psoriatic arthritis, ulcerative colitis) inhibits JAK1 and JAK3 and has been used off-label for alopecia areata, psoriasis, and vitiligo; ruxolitinib (approved for myelofibrosis, Polycythemia vera, GVHD) inhibits JAK1 and JAK 2 and has also been recently tested as a topical agent for vitiligo; and delgocitinib, inhibits all JAKs and was recently approved in Japan for the treatment of atopic dermatitis18.
Tapinarof is an aryl hydrocarbon receptor agonist that reduces oxidative stress, inhibits IL-4/IL-13 mediated STAT6 activation, and restores expression of filaggrin, loricrin and involucrin. It also increases IL-22 production and decreases IL-17A production (hence its efficacy shown in Phase 2 trials in psoriasis)19. Results from a 2019 JAAD-published Phase 2 randomized dose-finding study in atopic dermatitis showed promising results of 1% tapinarof cream compared to vehicle.
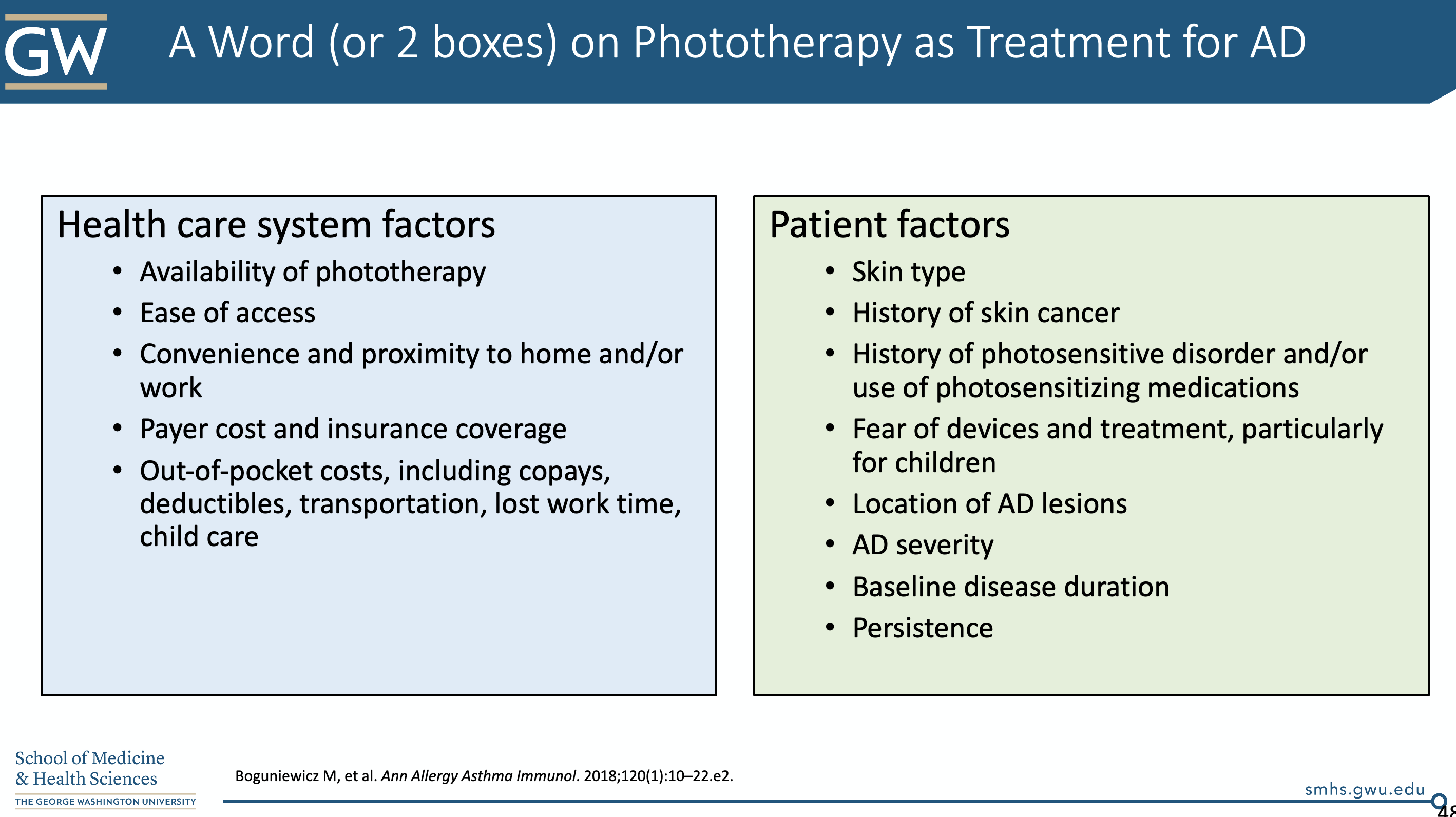
6. Phototherapy
Dr. Friedman briefly touched on this topic. Generally, we have to take several patient factors into account when considering phototherapy (notably NBUVB). We need to think about ease of access, payer cost/insurance coverage, skin type, history of photosensitivity, location of lesions, and others. We don’t typically start this during acute flares (it will make them worse).
7. Systemic treatment for atopic dermatitis
There are many (off-label) systemic treatments for atopic dermatitis. We do have to take into careful consideration potential side effects, as well as potential for teratogenicity in these agents. See the table below.

Take a look at Dr. Friedman’s golden slide below on other emerging topical and systemic therapies:
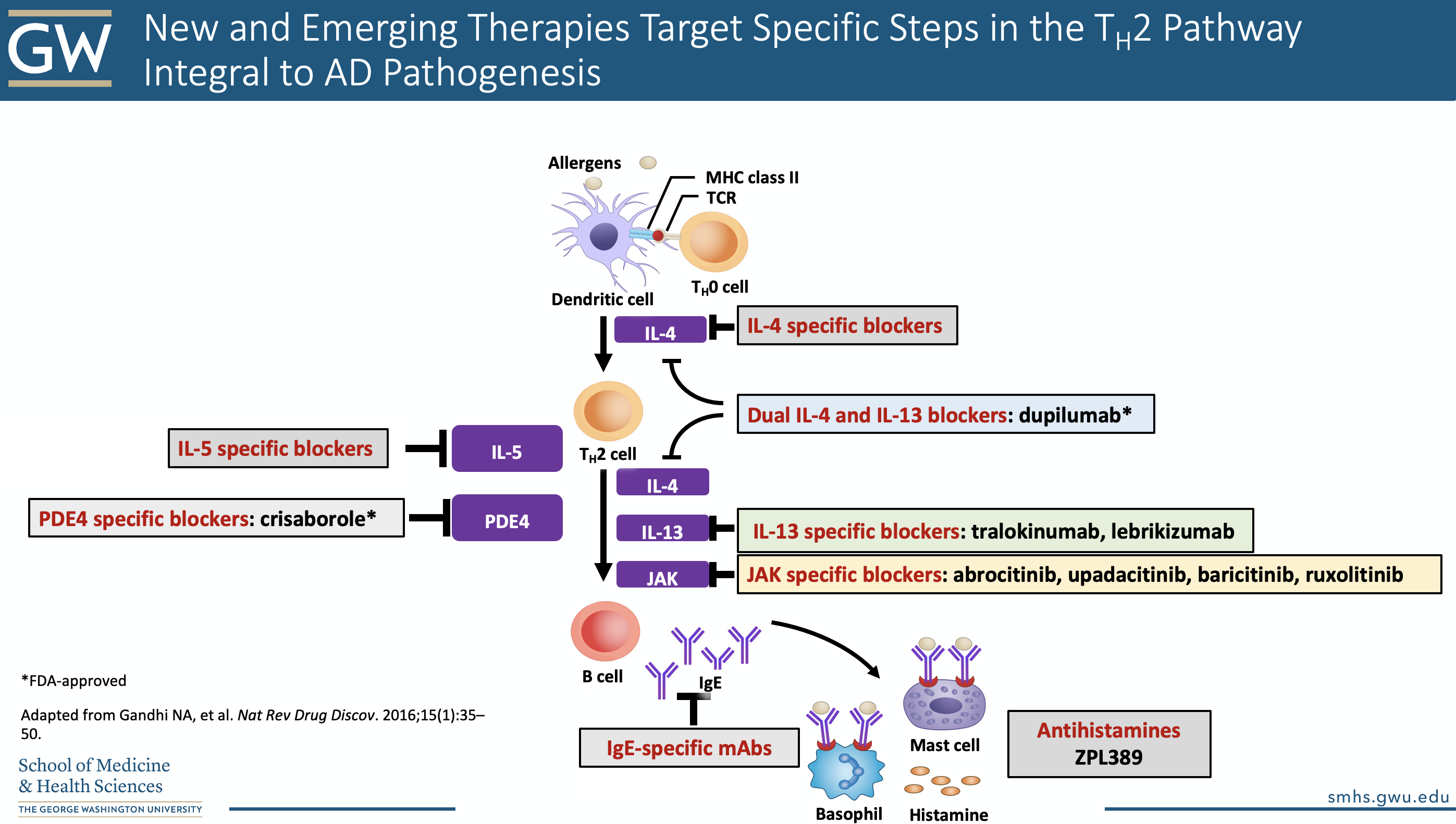
A note on upcoming oral JAK-inhibitors: Baricitinib, Abrocitinib, and Upadacitinib all appear to be effective in moderate-severe AD. They also have a rapid-onset of efficacy. The highest doses of Abrocitinib and Upadacitinib also appear to have greater efficacy than dupilumab, but head-to-head trials are pending. More data is needed to fully establish the safety profile of these drugs.
8. Dupixent: the ever-expanding label
Dupilumab is a fully human monoclonal antibody that inhibits the alpha subunit of the IL-4 receptor19. Binding of this receptor allows for dual antagonism of both IL-4 and IL-13 mediated inflammation (via the Th2 inflammatory pathway). This medication is approved for uncontrolled moderate-to-severe atopic dermatitis in ages 6 and above; moderate-to-severe asthma, and chronic rhinosinusitis with nasal polyposis. Dosing is available based on weight:
-
- ≥60 kg is an initial dose of 600 mg (two 300-mg pre-filled syringes) followed by 300 mg given every other week
- 30-60 kg is an initial dose of 400 mg (two 200-mg pre-filled syringes) followed by 200 mg given every other week
- 15-30 kg is an initial dose of 600 mg (two 300-mg pre-filled syringes) followed by 300 mg given every 4 weeks
Adverse events include20:
-
- Common (>10%) : antibody development, injection site reaction
- Somewhat common (1-10%): HSV infection, gastritis, arthralgia, conjunctivitis (about 10%), and others
- Rare (<1%): severe eye disease (punctal stenosis, cicatricial ectropion), inflammatory arthritis, alopecia areata, worsening facial AD/rosacea-like dermatitis, eosinophilic conditions, others.
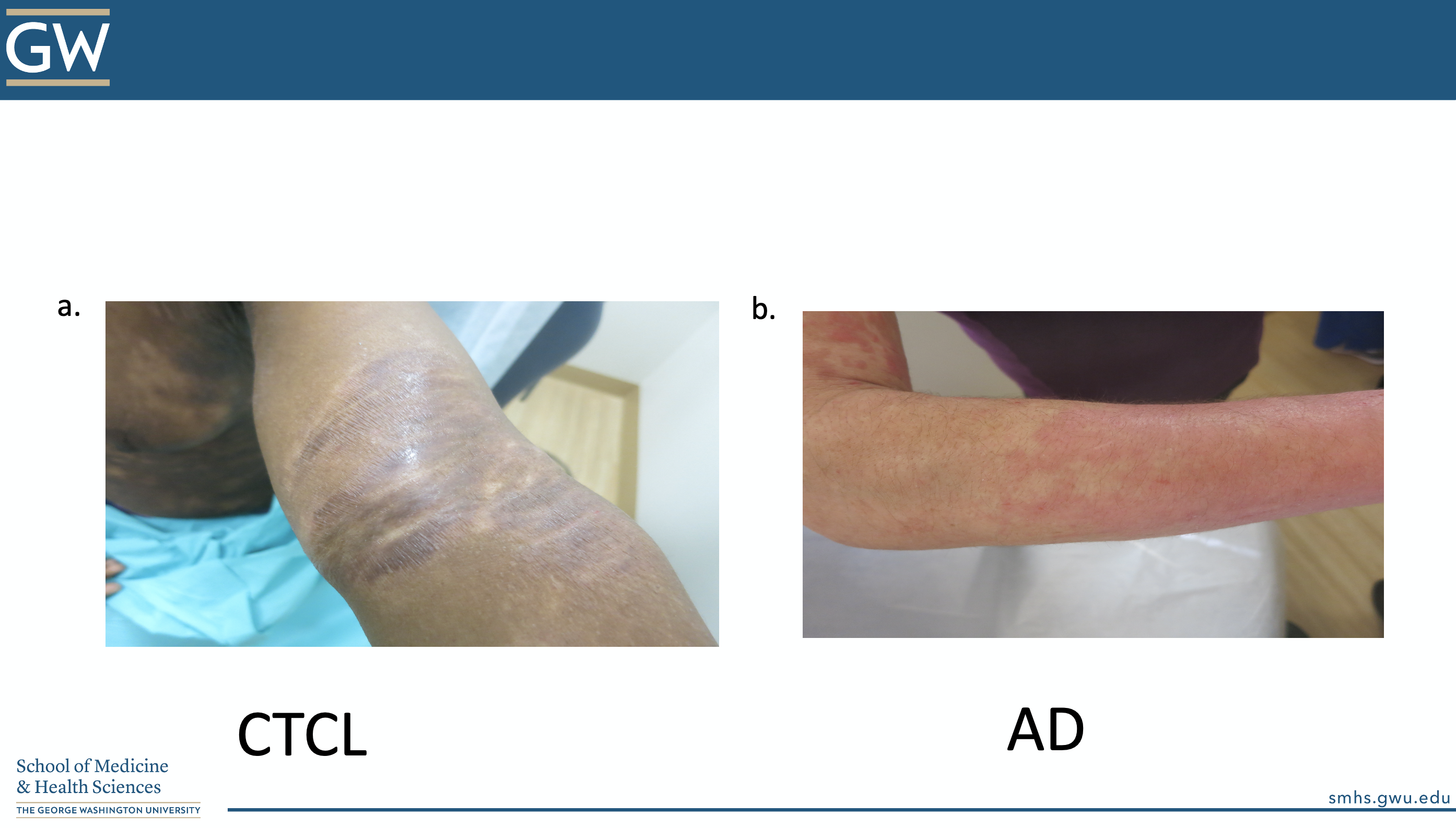
The ODAC Inflammatory Diseases Symposium was a real treat, with Dr. Friedman’s lecture, the cherry on top. Let’s end with a POP QUIZ!
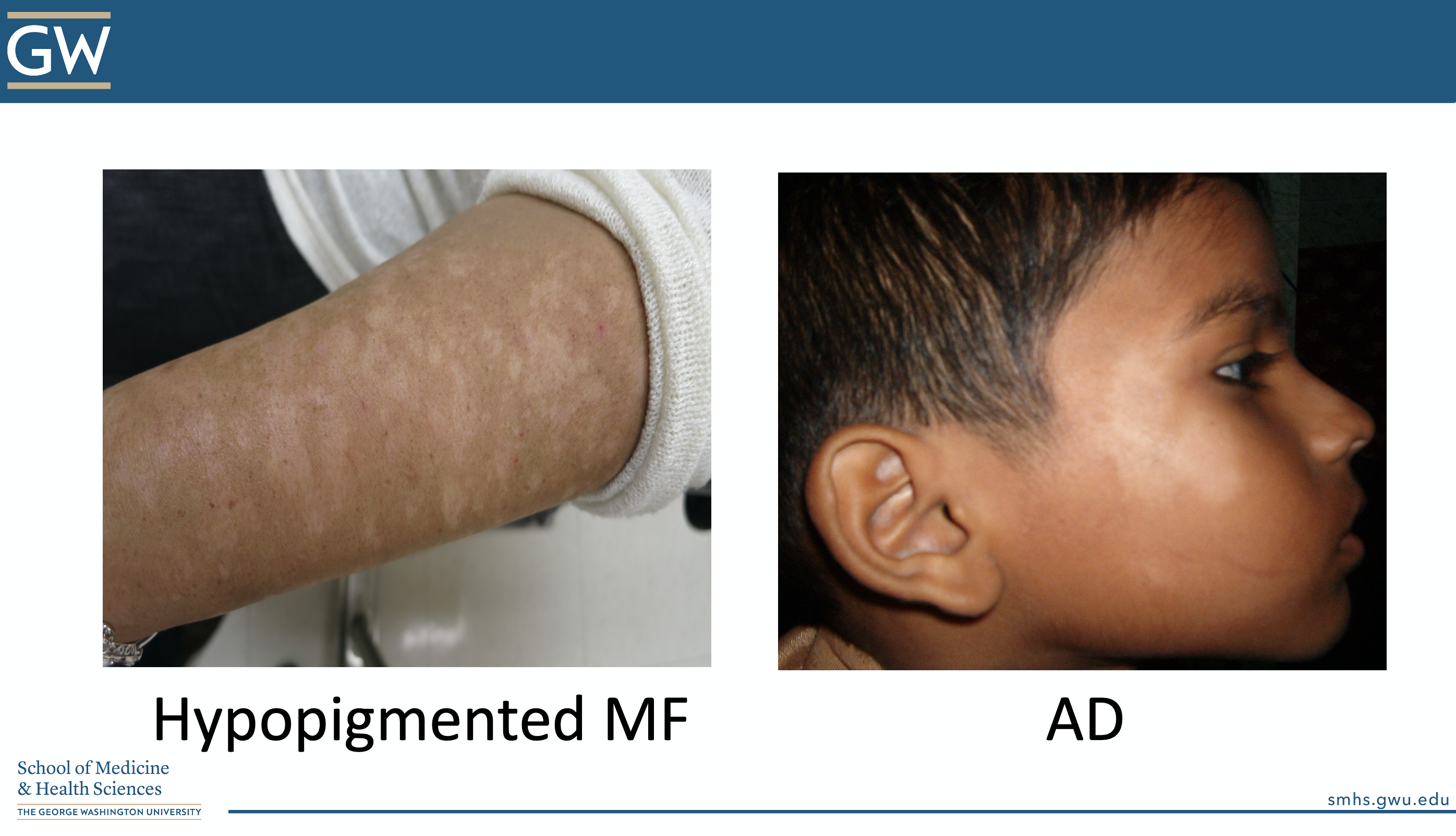
“Is this atopic dermatitis or NOT?”
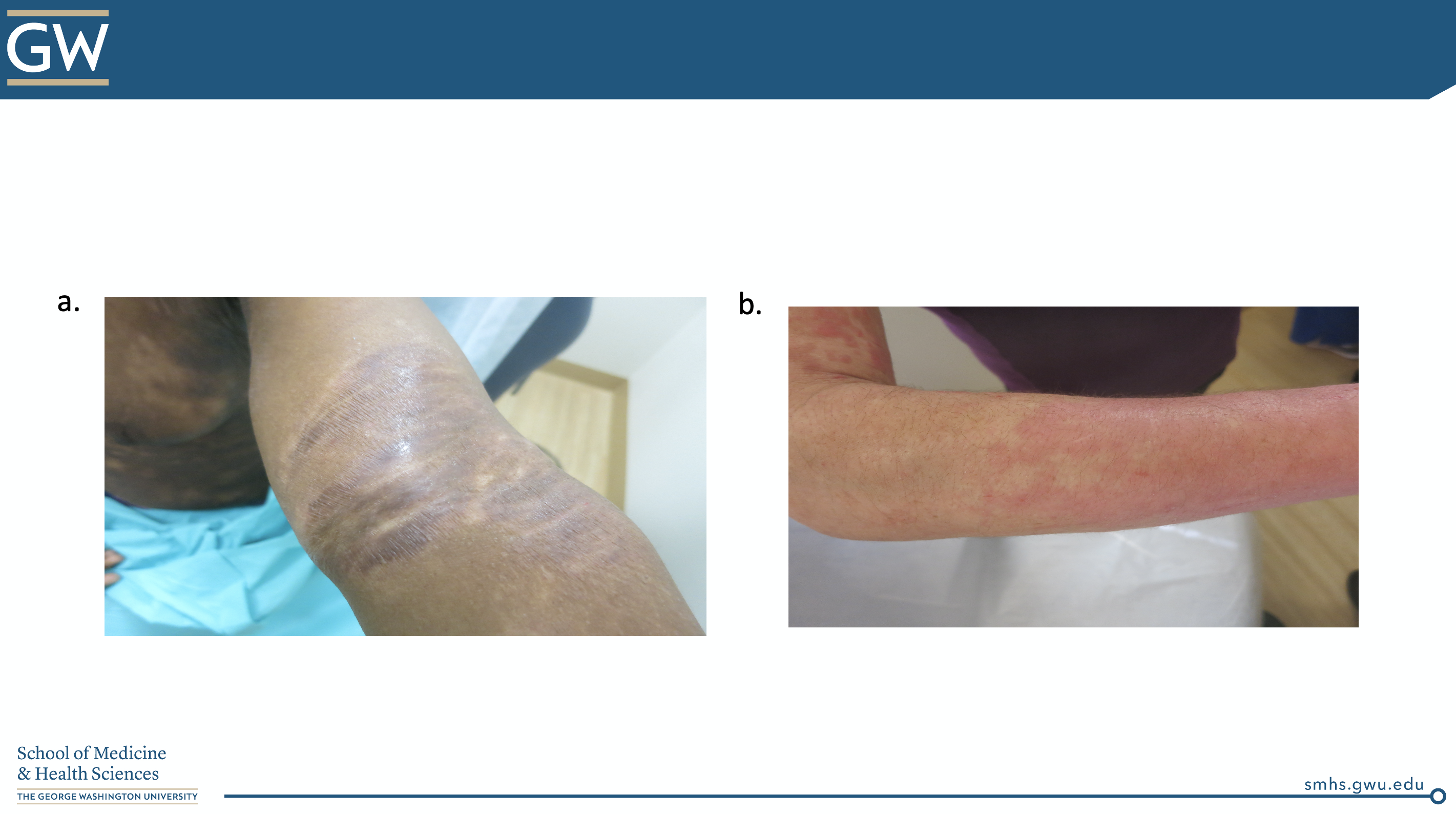
CORRECT ANSWER:
How often do you provide specific recommendations for soaps, bathing, or moisturizers to your patients? Do you have a favorite hand out or educational tool?
Please share your comments at the end of this post and on Instagram @nextstepsinderm!
This information was presented by Dr. Adam Friedman at the 2021 ODAC Sneak Peek Inflammatory Diseases Symposium held on December 3rd, 2020. The above highlights from her lecture were written and compiled by Jacqueline McKesey, MD, MS, Dermatology Resident at the University of Texas Southwestern Medical Center in Dallas, TX
Photo Credit: all clinical images and slides courtesy of Adam Friedman, MD
References
-
- Nakamura M, Abrouk M, Zhu H, Farahnik B, Koo J, Bhutani T. Update on the Systemic Risks of Superpotent Topical Steroids. J Drugs Dermatol. 2017 Jul 1;16(7):643-648. PMID: 28697215.
- Eczema Stats. National Eczema Association website. Accessed December 30, 2020. https://nationaleczema.org/research/eczema-facts/
- Silverberg JI, Simpson EL. Association between severe eczema in children and multiple comorbid conditions and increased healthcare utilization. Pediatr Allergy Immunol. 2013 Aug;24(5):476-86. doi: 10.1111/pai.12095. Epub 2013 Jun 16. PMID: 23773154; PMCID: PMC4397968.
- Thorsteinsdottir S, Stokholm J, Thyssen JP, et al. Genetic, Clinical, and Environmental Factors Associated With Persistent Atopic Dermatitis in Childhood. JAMA Dermatol.2019;155(1):50–57. doi:10.1001/jamadermatol.2018.4061
- Silverberg JI, Gelfand JM, Margolis DJ, Boguniewicz M, Fonacier L, Grayson MH, Ong PY, Chiesa Fuxench Z, Simpson EL. Atopic Dermatitis in US Adults: From Population to Health Care Utilization. J Allergy Clin Immunol Pract. 2019 May-Jun;7(5):1524-1532.e2. doi: 10.1016/j.jaip.2019.01.005. Epub 2019 Jan 14. PMID: 30654197.
- Eichenfield LF, Tom WL, Chamlin SL, Feldman SR, Hanifin JM, Simpson EL, Berger TG, Bergman JN, Cohen DE, Cooper KD, Cordoro KM, Davis DM, Krol A, Margolis DJ, Paller AS, Schwarzenberger K, Silverman RA, Williams HC, Elmets CA, Block J, Harrod CG, Smith Begolka W, Sidbury R. Guidelines of care for the management of atopic dermatitis: section 1. Diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014 Feb;70(2):338-51. doi: 10.1016/j.jaad.2013.10.010. Epub 2013 Nov 27. PMID: 24290431; PMCID: PMC4410183.
- Eichenfield LF, Tom WL, Berger TG, Krol A, Paller AS, Schwarzenberger K, Bergman JN, Chamlin SL, Cohen DE, Cooper KD, Cordoro KM, Davis DM, Feldman SR, Hanifin JM, Margolis DJ, Silverman RA, Simpson EL, Williams HC, Elmets CA, Block J, Harrod CG, Smith Begolka W, Sidbury R. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014 Jul;71(1):116-32. doi: 10.1016/j.jaad.2014.03.023. Epub 2014 May 9. PMID: 24813302; PMCID: PMC4326095.
- Sidbury R, Davis DM, Cohen DE, Cordoro KM, Berger TG, Bergman JN, Chamlin SL, Cooper KD, Feldman SR, Hanifin JM, Krol A, Margolis DJ, Paller AS, Schwarzenberger K, Silverman RA, Simpson EL, Tom WL, Williams HC, Elmets CA, Block J, Harrod CG, Begolka WS, Eichenfield LF; American Academy of Dermatology. Guidelines of care for the management of atopic dermatitis: section 3. Management and treatment with phototherapy and systemic agents. J Am Acad Dermatol. 2014 Aug;71(2):327-49. doi: 10.1016/j.jaad.2014.03.030. Epub 2014 May 9. PMID: 24813298; PMCID: PMC4410179.
- Sidbury R, Tom WL, Bergman JN, Cooper KD, Silverman RA, Berger TG, Chamlin SL, Cohen DE, Cordoro KM, Davis DM, Feldman SR, Hanifin JM, Krol A, Margolis DJ, Paller AS, Schwarzenberger K, Simpson EL, Williams HC, Elmets CA, Block J, Harrod CG, Smith Begolka W, Eichenfield LF. Guidelines of care for the management of atopic dermatitis: Section 4. Prevention of disease flares and use of adjunctive therapies and approaches. J Am Acad Dermatol. 2014 Dec;71(6):1218-33. doi: 10.1016/j.jaad.2014.08.038. Epub 2014 Sep 26. PMID: 25264237; PMCID: PMC4430554.
- Boguniewicz M, Fonacier L, Guttman-Yassky E, Ong PY, Silverberg J, Farrar JR. Atopic dermatitis yardstick: Practical recommendations for an evolving therapeutic landscape. Ann Allergy Asthma Immunol. 2018 Jan;120(1):10-22.e2. doi: 10.1016/j.anai.2017.10.039. PMID: 29273118.
- Simpson EL, Chalmers JR, Hanifin JM, Thomas KS, Cork MJ, McLean WH, Brown SJ, Chen Z, Chen Y, Williams HC. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014 Oct;134(4):818-23. doi: 10.1016/j.jaci.2014.08.005. PMID: 25282563; PMCID: PMC4180007.
- Horimukai K, Morita K, Narita M, Kondo M, Kitazawa H, Nozaki M, Shigematsu Y, Yoshida K, Niizeki H, Motomura K, Sago H, Takimoto T, Inoue E, Kamemura N, Kido H, Hisatsune J, Sugai M, Murota H, Katayama I, Sasaki T, Amagai M, Morita H, Matsuda A, Matsumoto K, Saito H, Ohya Y. Application of moisturizer to neonates prevents development of atopic dermatitis. J Allergy Clin Immunol. 2014 Oct;134(4):824-830.e6. doi: 10.1016/j.jaci.2014.07.060. PMID: 25282564.
- Czarnowicki T, Malajian D, Khattri S, Correa da Rosa J, Dutt R, Finney R, Dhingra N, Xiangyu P, Xu H, Estrada YD, Zheng X, Gilleaudeau P, Sullivan-Whalen M, Suaréz-Fariñas M, Shemer A, Krueger JG, Guttman-Yassky E. Petrolatum: Barrier repair and antimicrobial responses underlying this “inert” moisturizer. J Allergy Clin Immunol. 2016 Apr;137(4):1091-1102.e7. doi: 10.1016/j.jaci.2015.08.013. Epub 2015 Oct 1. PMID: 26431582.
- Allais B, Friedman A. ARTICLE: Colloidal Oatmeal Part I: History, Basic Science, Mechanism of Action, and Clinical Efficacy in the Treatment of Atopic Dermatitis. J Drugs Dermatol. 2020 Oct 1;19(10):s4-s7. PMID: 33026768.
- Seite S, Flores GE, Henley JB, Martin R, Zelenkova H, Aguilar L, Fierer N. Microbiome of affected and unaffected skin of patients with atopic dermatitis before and after emollient treatment. J Drugs Dermatol. 2014 Nov;13(11):1365-72. PMID: 25607704.
- Williams HC. Established corticosteroid creams should be applied only once daily in patients with atopic eczema. BMJ. 2007 Jun 16;334(7606):1272. doi: 10.1136/bmj.39195.636319.80. PMID: 17569936; PMCID: PMC1892503.
- Cotter DG, Schairer D, Eichenfield L. Emerging therapies for atopic dermatitis: JAK inhibitors. J Am Acad Dermatol. 2018 Mar;78(3 Suppl 1):S53-S62. doi: 10.1016/j.jaad.2017.12.019. Epub 2017 Dec 15. PMID: 29248518.
- Dhillon S. Delgocitinib: First Approval. Drugs. 2020 Apr;80(6):609-615. doi: 10.1007/s40265-020-01291-2. PMID: 32166597.
- Peppers J, Paller AS, Maeda-Chubachi T, Wu S, Robbins K, Gallagher K, Kraus JE. A phase 2, randomized dose-finding study of tapinarof (GSK2894512 cream) for the treatment of atopic dermatitis. J Am Acad Dermatol. 2019 Jan;80(1):89-98.e3. doi: 10.1016/j.jaad.2018.06.047. Epub 2018 Jul 3. PMID: 30554600.
- Dupixent (dupilumab) [prescribing information]. Tarrytown, NY: Regeneron Pharmaceuticals; November 2020.

