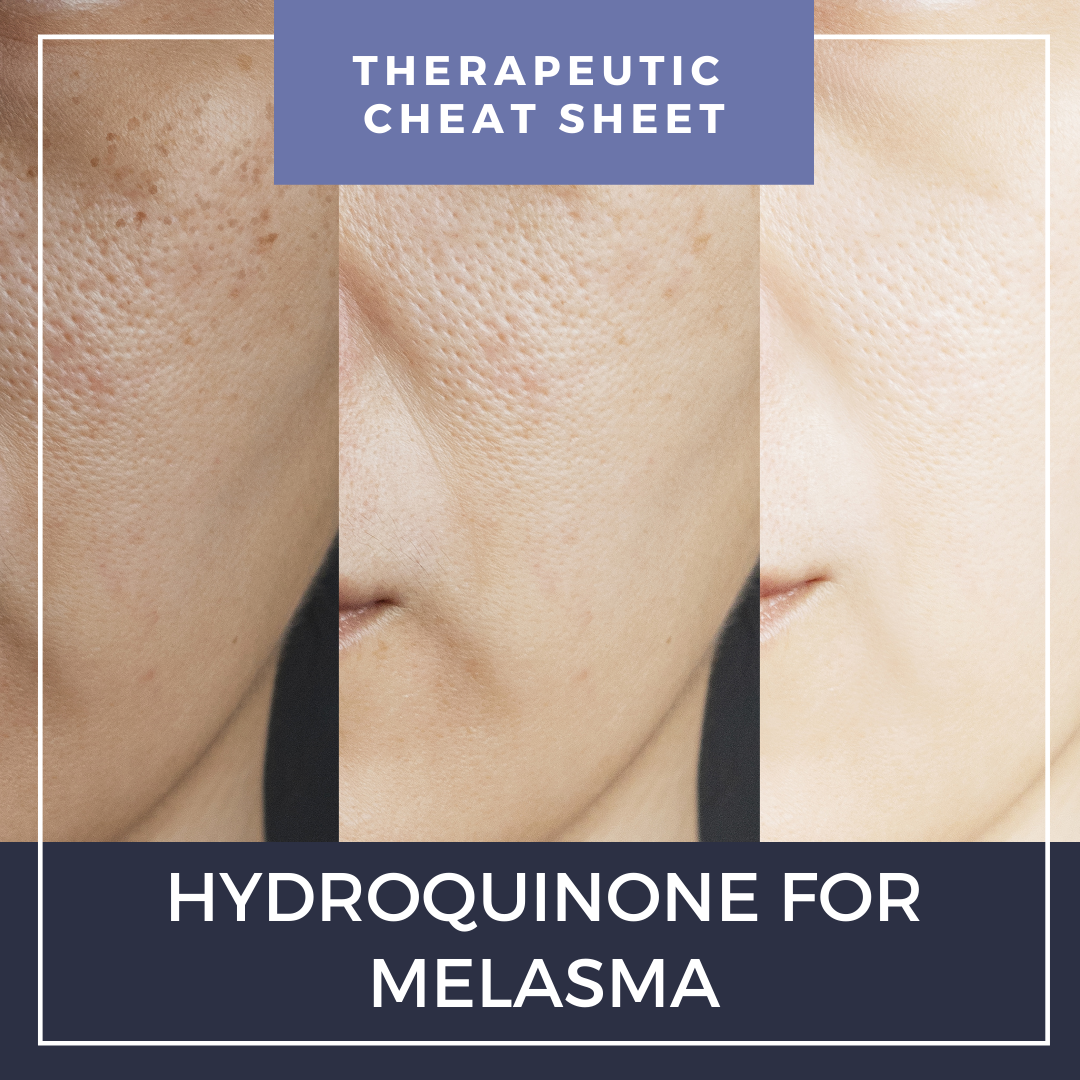
Hydroquinone is the most commonly used depigmentation agent and is a first-line treatment for melasma.1Hydroquinone was available previously in over-the-counter products and is currently available in prescription formulations with common concentration ranges of 0.4 to 5%. Prescription strengths available in the United States include hydroquinone 2% and 4% cream and hydroquinone 2% gel.2,3 In September 2020, the FDA enforced the CARES Act and Over-the-Counter Drug Monograph Reform, prohibiting the distribution of over-the-counter hydroquinone products.4 We continue our series, Therapeutic Cheat Sheet, with a closer look at hydroquinone 4% cream.
Hydroquinone Therapeutic Cheat Sheet
Compiled by: Kamaria Nelson, MD | Reviewed by: Adam Friedman, MD
TRADE NAME
-
- Hydroquinone 4% cream
HYDROQUINONE MECHANISM OF ACTION
-
- Hydroquinone or 1,4-dihydroxybenzene is a derivative of phenol and metabolized to glutathione.5Through topical application of hydroquinone, there is reversible depigmentation of the skin via inhibition of tyrosinase which normally converts tyrosine to 3,4-dihydroxypheylalanine (dopa).5,6, 7 Additionally, there is suppression of other melanocyte metabolic processes decreasing melanin formation via selective damage to melanosomes and melanocytes through production of reactive oxygen radicals.6,7,8
FDA-APPROVED USE
-
- Hydroquinone is currently only FDA-approved in combination with fluocinolone acetonide 0.01% and tretinoin 0.05% cream under the trade name TRI-LUMA for moderate to severe melasma of the face.9
OFF-LABEL USES OF HYDROQUINONE
-
- Disorders of hyperpigmentation including but not limited to chloasma, melasma, ephelides, and solar lentigines 5,6,8
HYDROQUINONE DOSING
-
- Topical application to affected areas twice a day.6 If there is no improvement in 2 months, use of product should be discontinued.6
- Efficacy improves with use of daily sunscreen.10
- There is no standardized treatment duration, but most studies utilized twice a day dosing for 12 to 24 weeks duration.10
SIDE EFFECTS ASSOCIATED WITH HYDROQUINONE
-
- Mild erythema
- Burning sensation
- Dryness
- Fissuring of paranasal and infraorbital areas
- Stinging
- Localized hypersensitivity
- Pruritus
- Scaling
- Exogenous ochronosis
HYDROQUINONE WARNINGS
-
- Contains sodium metabisulfite which may cause an allergic-type reaction including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in susceptible people.
- An effective broad-spectrum sun blocking agent should be used and unnecessary solar exposure avoided.
- Hydroquinone may produce exogenous ochronosis, a gradual blue-black darkening of the skin. If this occurs, discontinue treatment.
HYDROQUINONE CONTRAINDICATIONS
-
- Prior history of sensitivity or allergic reaction to hydroquinone or any other ingredients in the product.
PREGNANCY
-
- There are no studies or adequate data on risk associated in pregnant women. Animal reproduction studies have not been conducted with topical hydroquinone.
- No data on the presence of hydroquinone in human milk, effects on milk production, or effects on infants who are breastfed.
MONITORING
-
- No recommended monitoring guidelines
CLICK ON THE IMAGE BELOW TO ENLARGE AND/OR DOWNLOAD YOUR THERAPEUTIC CHEAT SHEET
Overall, hydroquinone appears to be a safe and effective therapy for many disorders of hyperpigmentation with greatest utility in treating melasma. Practitioners should continue to take note of newly published reports concerning the safety of this therapeutic option in the diversity of clinical settings in which it is utilized. Hydroquinone-induced exogenous ochronosis is a potential side effect which when present, is difficult to treat. The International Journal of Dermatology published a systematic review of the risk factors of patients with exogenous ochronosis due to hydroquinone as highlighted below. It was found that higher concentrations and longer treatment courses were most commonly reported with the onset of exogenous ochronosis.
Stephanie Ishack,PhD and Shari R. Lipner, MD, PhD
Abstract:
Exogenous ochronosis is a potential side effect associated with hydroquinone, and treatment is often unsatisfactory. Our study objectives were to review data on hydroquinone-associated ochronosis to determine risk factors for patients experiencing this adverse event. On September 27, 2020 (MEDLINE/PubMed), and October 30, 2020 (Scopus and Web of Science), databases were searched for “ochronosis + hydroquinone” by both authors to reduce risk basis. PRISMA reporting guidelines were used to select 56 articles with a total of 126 patients with hydroquinone-associated ochronosis. Included articles described hydroquinone-associated ochronosis. Articles were excluded if they had irrelevant content, were non-English language text, and were non-case studies. Full text articles were assessed and recorded. Cross-tabulation analysis was performed on categorical data, and Fisher exact test was performed. Ochronosis was most often reported in middle-aged women (53.2%), of African descent (45.2%), Black races (55.5%), and Fitzpatrick skin types V-VI (52.4%). It was most frequently reported with unknown and hydroquinone concentrations greater than 4% (32.5 and 35.7% cases, respectively). Median duration of use was 5 years, with only four cases reported with courses 3 months or shorter and eight cases reported with use 1 year or less. All patients presented with facial blue-black or gray-blue macules in a reticulate, lace-like fashion. Histopathology consistently showed solar elastosis and brownish-yellow, ‘banana-shaped’ fibers between degenerated collagen fibers of the papillary dermis. Based on these findings, we conclude that hydroquinone in concentrations above 4% and in treatment courses longer than 3 months may be associated with new-onset ochronosis. Read the full article here.
References
-
- Zubair R, Lyons AB, Vellaichamy G, Peacock A, Hamzavi I. What’s New in Pigmentary Disorders. Dermatol Clin. 2019 Apr;37(2):175-181. doi: 10.1016/j.det.2018.12.008. Epub 2019 Feb 16. PMID: 30850040.
- (2021, March 3). Lustra, Neostrata (hydroquinone) dosing, indications, interactions, adverse effects, and more. Retrieved January 30, 2022, from https://reference.medscape.com/drug/lustra-melquin-hydroquinone-343660
- Epocrates (2021). Hydroquinone topical. hydroquinone topical Entire Monograph – Epocrates Web. Retrieved January 30, 2022, from https://online.dev.epocrates.com/drugs/3710/hydroquinone-topical/Monograph
- Ishack S, Lipner SR. Exogenous ochronosis associated with hydroquinone: a systematic review. Int J Dermatol. 2021 Sep 6. doi: 10.1111/ijd.15878. Epub ahead of print. PMID: 34486734.
- Searle T, Al-Niaimi F, Ali FR. Hydroquinone: myths and reality. Clin Exp Dermatol. 2021 Jun;46(4):636-640. doi: 10.1111/ced.14480. Epub 2020 Nov 7. PMID: 33159818.
- National Institutes of Health. DailyMed – hydroquinone 4%- hydroquinone cream. U.S. National Library of Medicine. Retrieved January 30, 2022, from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=595dfc50-ee6f-41b5-ab5b-4c293aee342b
- Andersen FA, Bergfeld WF, Belsito DV, Hill RA, Klaassen CD, Liebler DC, Marks JG Jr, Shank RC, Slaga TJ, Snyder PW. Final amended safety assessment of hydroquinone as used in cosmetics. Int J Toxicol. 2010 Nov-Dec;29(6 Suppl):274S-87. doi: 10.1177/1091581810385957. PMID: 21164074.
- Bolognia J, Schaffer JV, Cerroni L. Dermatology. Fourth edition. Elsevier; 2018.
- Galderma: TRI-LUMA ® Cream (fluocinolone acetonide 0.01%, hydroquinone 4%, tretinoin 0.05%). Package Insert. US Prescribing Information and Patient Information. Revised 2013 Available at https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/021112s006lbl.pdf
- McKesey J, Tovar-Garza A, Pandya AG. Melasma Treatment: An Evidence-Based Review. Am J Clin Dermatol. 2020 Apr;21(2):173-225. doi: 10.1007/s40257-019-00488-w. PMID: 31802394.
- Neagu N, Conforti C, Agozzino M, Marangi GF, Morariu SH, Pellacani G, Persichetti P, Piccolo D, Segreto F, Zalaudek I, Dianzani C. Melasma treatment: a systematic review. J Dermatolog Treat. 2021 Apr 14:1-39. doi: 10.1080/09546634.2021.1914313. Epub ahead of print. PMID: 33849384.